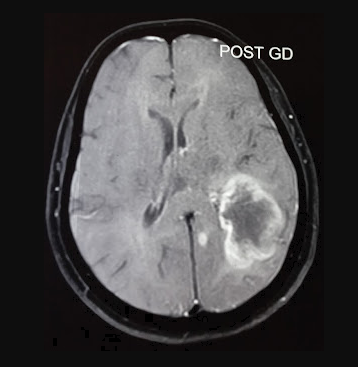
[1]
Li C, Li B, Wang H, Qu L, Liu H, Weng C, Han J, Li Y. Role of N6-methyladenosine methylation in glioma: recent insights and future directions. Cellular & molecular biology letters. 2023 Dec 11:28(1):103. doi: 10.1186/s11658-023-00514-0. Epub 2023 Dec 11
[PubMed PMID: 38072944]
Level 3 (low-level) evidence
[2]
Thakkar JP, Dolecek TA, Horbinski C, Ostrom QT, Lightner DD, Barnholtz-Sloan JS, Villano JL. Epidemiologic and molecular prognostic review of glioblastoma. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2014 Oct:23(10):1985-96. doi: 10.1158/1055-9965.EPI-14-0275. Epub 2014 Jul 22
[PubMed PMID: 25053711]
[3]
Ostrom QT, Gittleman H, Xu J, Kromer C, Wolinsky Y, Kruchko C, Barnholtz-Sloan JS. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2009-2013. Neuro-oncology. 2016 Oct 1:18(suppl_5):v1-v75. doi: 10.1093/neuonc/now207. Epub
[PubMed PMID: 28475809]
[4]
Gilard V, Tebani A, Dabaj I, Laquerrière A, Fontanilles M, Derrey S, Marret S, Bekri S. Diagnosis and Management of Glioblastoma: A Comprehensive Perspective. Journal of personalized medicine. 2021 Apr 1:11(4):. doi: 10.3390/jpm11040258. Epub 2021 Apr 1
[PubMed PMID: 33915852]
Level 3 (low-level) evidence
[5]
Tan AC, Ashley DM, López GY, Malinzak M, Friedman HS, Khasraw M. Management of glioblastoma: State of the art and future directions. CA: a cancer journal for clinicians. 2020 Jul:70(4):299-312. doi: 10.3322/caac.21613. Epub 2020 Jun 1
[PubMed PMID: 32478924]
Level 3 (low-level) evidence
[6]
Rong L, Li N, Zhang Z. Emerging therapies for glioblastoma: current state and future directions. Journal of experimental & clinical cancer research : CR. 2022 Apr 15:41(1):142. doi: 10.1186/s13046-022-02349-7. Epub 2022 Apr 15
[PubMed PMID: 35428347]
Level 3 (low-level) evidence
[7]
Hodges LC, Smith JL, Garrett A, Tate S. Prevalence of glioblastoma multiforme in subjects with prior therapeutic radiation. The Journal of neuroscience nursing : journal of the American Association of Neuroscience Nurses. 1992 Apr:24(2):79-83
[PubMed PMID: 1318344]
[8]
Brenner AV, Linet MS, Fine HA, Shapiro WR, Selker RG, Black PM, Inskip PD. History of allergies and autoimmune diseases and risk of brain tumors in adults. International journal of cancer. 2002 May 10:99(2):252-9
[PubMed PMID: 11979441]
[9]
Scheurer ME, Amirian ES, Davlin SL, Rice T, Wrensch M, Bondy ML. Effects of antihistamine and anti-inflammatory medication use on risk of specific glioma histologies. International journal of cancer. 2011 Nov 1:129(9):2290-6. doi: 10.1002/ijc.25883. Epub 2011 Apr 4
[PubMed PMID: 21190193]
[10]
Hochberg F, Toniolo P, Cole P, Salcman M. Nonoccupational risk indicators of glioblastoma in adults. Journal of neuro-oncology. 1990 Feb:8(1):55-60
[PubMed PMID: 2319291]
[11]
Urbańska K, Sokołowska J, Szmidt M, Sysa P. Glioblastoma multiforme - an overview. Contemporary oncology (Poznan, Poland). 2014:18(5):307-12. doi: 10.5114/wo.2014.40559. Epub 2014 Aug 4
[PubMed PMID: 25477751]
Level 3 (low-level) evidence
[12]
Tebha SS, Ali Memon S, Mehmood Q, Mukherjee D, Abdi H, Negida A. Glioblastoma management in low and middle-income countries; existing challenges and policy recommendations. Brain & spine. 2023:3():101775. doi: 10.1016/j.bas.2023.101775. Epub 2023 Jul 8
[PubMed PMID: 38021027]
[13]
Slika H, Karimov Z, Alimonti P, Abou-Mrad T, De Fazio E, Alomari S, Tyler B. Preclinical Models and Technologies in Glioblastoma Research: Evolution, Current State, and Future Avenues. International journal of molecular sciences. 2023 Nov 14:24(22):. doi: 10.3390/ijms242216316. Epub 2023 Nov 14
[PubMed PMID: 38003507]
[14]
Grochans S, Cybulska AM, Simińska D, Korbecki J, Kojder K, Chlubek D, Baranowska-Bosiacka I. Epidemiology of Glioblastoma Multiforme-Literature Review. Cancers. 2022 May 13:14(10):. doi: 10.3390/cancers14102412. Epub 2022 May 13
[PubMed PMID: 35626018]
[15]
Ohgaki H, Dessen P, Jourde B, Horstmann S, Nishikawa T, Di Patre PL, Burkhard C, Schüler D, Probst-Hensch NM, Maiorka PC, Baeza N, Pisani P, Yonekawa Y, Yasargil MG, Lütolf UM, Kleihues P. Genetic pathways to glioblastoma: a population-based study. Cancer research. 2004 Oct 1:64(19):6892-9
[PubMed PMID: 15466178]
[16]
Jiang C, Mogilevsky C, Belal Z, Kurtz G, Alonso-Basanta M. Hypofractionation in Glioblastoma: An Overview of Palliative, Definitive, and Exploratory Uses. Cancers. 2023 Nov 29:15(23):. doi: 10.3390/cancers15235650. Epub 2023 Nov 29
[PubMed PMID: 38067354]
Level 3 (low-level) evidence
[17]
Tiwari S, Han Z. Immunotherapy: Advancing glioblastoma treatment-A narrative review of scientific studies. Cancer reports (Hoboken, N.J.). 2023 Dec 9:7(2):e1947. doi: 10.1002/cnr2.1947. Epub 2023 Dec 9
[PubMed PMID: 38069593]
Level 3 (low-level) evidence
[18]
Agosti E, Zeppieri M, De Maria L, Tedeschi C, Fontanella MM, Panciani PP, Ius T. Glioblastoma Immunotherapy: A Systematic Review of the Present Strategies and Prospects for Advancements. International journal of molecular sciences. 2023 Oct 10:24(20):. doi: 10.3390/ijms242015037. Epub 2023 Oct 10
[PubMed PMID: 37894718]
Level 1 (high-level) evidence
[19]
Olar A, Aldape KD. Using the molecular classification of glioblastoma to inform personalized treatment. The Journal of pathology. 2014 Jan:232(2):165-77. doi: 10.1002/path.4282. Epub
[PubMed PMID: 24114756]
[20]
Stoyanov GS, Dzhenkov D, Ghenev P, Iliev B, Enchev Y, Tonchev AB. Cell biology of glioblastoma multiforme: from basic science to diagnosis and treatment. Medical oncology (Northwood, London, England). 2018 Jan 31:35(3):27. doi: 10.1007/s12032-018-1083-x. Epub 2018 Jan 31
[PubMed PMID: 29387965]
[21]
Jovčevska I. Genetic secrets of long-term glioblastoma survivors. Bosnian journal of basic medical sciences. 2019 May 20:19(2):116-124. doi: 10.17305/bjbms.2018.3717. Epub 2019 May 20
[PubMed PMID: 30114377]
[22]
Linkous AG, Yazlovitskaya EM. Angiogenesis in glioblastoma multiforme: navigating the maze. Anti-cancer agents in medicinal chemistry. 2011 Oct:11(8):712-8
[PubMed PMID: 21707499]
[23]
Stoyanov GS, Petkova L, Dzhenkov DL. A Practical Approach to the Differential Diagnosis of Intracranial Tumors: Gross, Histology, and Immunoprofile-based Algorithm. Cureus. 2019 Dec 15:11(12):e6384. doi: 10.7759/cureus.6384. Epub 2019 Dec 15
[PubMed PMID: 31938662]
[24]
Katsetos CD, Dráberová E, Legido A, Dumontet C, Dráber P. Tubulin targets in the pathobiology and therapy of glioblastoma multiforme. I. Class III beta-tubulin. Journal of cellular physiology. 2009 Dec:221(3):505-13. doi: 10.1002/jcp.21870. Epub
[PubMed PMID: 19650075]
[25]
Schultz S, Pinsky GS, Wu NC, Chamberlain MC, Rodrigo AS, Martin SE. Fine needle aspiration diagnosis of extracranial glioblastoma multiforme: Case report and review of the literature. CytoJournal. 2005 Nov 14:2():19
[PubMed PMID: 16287502]
Level 3 (low-level) evidence
[26]
Kaina B. Temozolomide, Procarbazine and Nitrosoureas in the Therapy of Malignant Gliomas: Update of Mechanisms, Drug Resistance and Therapeutic Implications. Journal of clinical medicine. 2023 Nov 30:12(23):. doi: 10.3390/jcm12237442. Epub 2023 Nov 30
[PubMed PMID: 38068493]
[27]
Ndirangu B, Bryan K, Nduom E. Extent of Resection and Outcomes of Patients with Primary Malignant Brain Tumors. Current treatment options in oncology. 2023 Dec:24(12):1948-1961. doi: 10.1007/s11864-023-01158-0. Epub 2023 Dec 13
[PubMed PMID: 38091186]
[28]
Wang L, Liang B, Li YI, Liu X, Huang J, Li YM. What is the advance of extent of resection in glioblastoma surgical treatment-a systematic review. Chinese neurosurgical journal. 2019:5():2. doi: 10.1186/s41016-018-0150-7. Epub 2019 Feb 1
[PubMed PMID: 32922902]
Level 1 (high-level) evidence
[29]
Revilla-Pacheco F, Rodríguez-Salgado P, Barrera-Ramírez M, Morales-Ruiz MP, Loyo-Varela M, Rubalcava-Ortega J, Herrada-Pineda T. Extent of resection and survival in patients with glioblastoma multiforme: Systematic review and meta-analysis. Medicine. 2021 Jun 25:100(25):e26432. doi: 10.1097/MD.0000000000026432. Epub
[PubMed PMID: 34160432]
Level 1 (high-level) evidence
[30]
Chojak R, Koźba-Gosztyła M, Słychan K, Gajos D, Kotas M, Tyliszczak M, Czapiga B. Impact of surgical resection of butterfly glioblastoma on survival: a meta-analysis based on comparative studies. Scientific reports. 2021 Jul 6:11(1):13934. doi: 10.1038/s41598-021-93441-z. Epub 2021 Jul 6
[PubMed PMID: 34230597]
Level 2 (mid-level) evidence
[31]
Bonosi L, Marrone S, Benigno UE, Buscemi F, Musso S, Porzio M, Silven MP, Torregrossa F, Grasso G. Maximal Safe Resection in Glioblastoma Surgery: A Systematic Review of Advanced Intraoperative Image-Guided Techniques. Brain sciences. 2023 Jan 28:13(2):. doi: 10.3390/brainsci13020216. Epub 2023 Jan 28
[PubMed PMID: 36831759]
Level 1 (high-level) evidence
[32]
Angom RS, Nakka NMR, Bhattacharya S. Advances in Glioblastoma Therapy: An Update on Current Approaches. Brain sciences. 2023 Oct 31:13(11):. doi: 10.3390/brainsci13111536. Epub 2023 Oct 31
[PubMed PMID: 38002496]
Level 3 (low-level) evidence
[33]
Zhang JJY, Lee KS, Voisin MR, Hervey-Jumper SL, Berger MS, Zadeh G. Awake craniotomy for resection of supratentorial glioblastoma: a systematic review and meta-analysis. Neuro-oncology advances. 2020 Jan-Dec:2(1):vdaa111. doi: 10.1093/noajnl/vdaa111. Epub 2020 Sep 18
[PubMed PMID: 33063012]
Level 3 (low-level) evidence
[34]
Ramakrishnan PK, Saeed F, Thomson S, Corns R, Mathew RK, Sivakumar G. Awake craniotomy for high-grade gliomas - a prospective cohort study in a UK tertiary-centre. The surgeon : journal of the Royal Colleges of Surgeons of Edinburgh and Ireland. 2024 Feb:22(1):e3-e12. doi: 10.1016/j.surge.2023.11.002. Epub 2023 Nov 25
[PubMed PMID: 38008681]
[35]
Prajapati HP, Singh DK. Recurrent glioblastoma in elderly: Options and decision for the treatment. Surgical neurology international. 2022:13():397. doi: 10.25259/SNI_552_2022. Epub 2022 Sep 2
[PubMed PMID: 36128156]
[36]
De Vleeschouwer S, Manrique-Guzmán S, Herrada-Pineda T, Revilla-Pacheco F. Surgical Management of Glioblastoma. Glioblastoma. 2017 Sep 27:():
[PubMed PMID: 29251865]
[37]
Domino JS, Ormond DR, Germano IM, Sami M, Ryken TC, Olson JJ. Cytoreductive surgery in the management of newly diagnosed glioblastoma in adults: a systematic review and evidence-based clinical practice guideline update. Journal of neuro-oncology. 2020 Nov:150(2):121-142. doi: 10.1007/s11060-020-03606-5. Epub 2020 Nov 19
[PubMed PMID: 33215341]
Level 1 (high-level) evidence
[38]
Xu S, Frakulli R, Lin Y. Comparison of the Effectiveness of Radiotherapy with 3D-CRT, IMRT, VMAT and PT for Newly Diagnosed Glioblastoma: A Bayesian Network Meta-Analysis. Cancers. 2023 Dec 3:15(23):. doi: 10.3390/cancers15235698. Epub 2023 Dec 3
[PubMed PMID: 38067401]
Level 1 (high-level) evidence
[39]
Colamaria A, Leone A, Fochi NP, Di Napoli V, Giordano G, Landriscina M, Patel K, Carbone F. Tumor treating fields for the treatment of glioblastoma: Current understanding and future perspectives. Surgical neurology international. 2023:14():394. doi: 10.25259/SNI_674_2023. Epub 2023 Nov 10
[PubMed PMID: 38053701]
Level 3 (low-level) evidence
[40]
Xiang X, Ji Z, Jin J. Brachytherapy is an effective and safe salvage option for re-irradiation in recurrent glioblastoma (rGBM): A systematic review. Radiotherapy and oncology : journal of the European Society for Therapeutic Radiology and Oncology. 2024 Jan:190():110012. doi: 10.1016/j.radonc.2023.110012. Epub 2023 Nov 14
[PubMed PMID: 37972737]
Level 1 (high-level) evidence
[41]
de Paula GA, de Paula MC, Dutra JAP, Carvalho SG, Di Filippo LD, Oliveira Villanova JC, Chorilli M. Targeted Polymeric Nanoparticles as a Strategy for the Treatment of Glioblastoma: A Review. Current drug delivery. 2023 Nov 27:():. doi: 10.2174/0115672018257713231107060630. Epub 2023 Nov 27
[PubMed PMID: 38013438]
[42]
Wahyuhadi J, Immadoel Haq IB, Arifianto MR, Sulistyono B, Meizikri R, Rosada A, Sigit Prakoeswa CR, Susilo RI. Active Immunotherapy for Glioblastoma Treatment: A Systematic Review and Meta-Analysis. Cancer control : journal of the Moffitt Cancer Center. 2022 Jan-Dec:29():10732748221079474. doi: 10.1177/10732748221079474. Epub
[PubMed PMID: 36748348]
Level 1 (high-level) evidence
[43]
Begagić E, Pugonja R, Bečulić H, Čeliković A, Tandir Lihić L, Kadić Vukas S, Čejvan L, Skomorac R, Selimović E, Jaganjac B, Juković-Bihorac F, Jusić A, Pojskić M. Molecular Targeted Therapies in Glioblastoma Multiforme: A Systematic Overview of Global Trends and Findings. Brain sciences. 2023 Nov 17:13(11):. doi: 10.3390/brainsci13111602. Epub 2023 Nov 17
[PubMed PMID: 38002561]
Level 1 (high-level) evidence
[44]
Wang JL, Scheitler KM, Wenger NM, Elder JB. Viral therapies for glioblastoma and high-grade gliomas in adults: a systematic review. Neurosurgical focus. 2021 Feb:50(2):E2. doi: 10.3171/2020.11.FOCUS20854. Epub
[PubMed PMID: 33524943]
Level 1 (high-level) evidence
[45]
Davidson CL, Vengoji R, Jain M, Batra SK, Shonka N. Biological, diagnostic and therapeutic implications of exosomes in glioma. Cancer letters. 2024 Feb 1:582():216592. doi: 10.1016/j.canlet.2023.216592. Epub 2023 Dec 12
[PubMed PMID: 38092145]
Level 3 (low-level) evidence
[46]
Lunavat TR, Nieland L, Vrijmoet AB, Zargani-Piccardi A, Samaha Y, Breyne K, Breakefield XO. Roles of extracellular vesicles in glioblastoma: foes, friends and informers. Frontiers in oncology. 2023:13():1291177. doi: 10.3389/fonc.2023.1291177. Epub 2023 Nov 24
[PubMed PMID: 38074665]
[47]
Gokden M. If it is Not a Glioblastoma, Then What is it? A Differential Diagnostic Review. Advances in anatomic pathology. 2017 Nov:24(6):379-391. doi: 10.1097/PAP.0000000000000170. Epub
[PubMed PMID: 28885262]
Level 3 (low-level) evidence
[48]
Walker MD, Strike TA, Sheline GE. An analysis of dose-effect relationship in the radiotherapy of malignant gliomas. International journal of radiation oncology, biology, physics. 1979 Oct:5(10):1725-31
[PubMed PMID: 231022]
[49]
Chang CH, Horton J, Schoenfeld D, Salazer O, Perez-Tamayo R, Kramer S, Weinstein A, Nelson JS, Tsukada Y. Comparison of postoperative radiotherapy and combined postoperative radiotherapy and chemotherapy in the multidisciplinary management of malignant gliomas. A joint Radiation Therapy Oncology Group and Eastern Cooperative Oncology Group study. Cancer. 1983 Sep 15:52(6):997-1007
[PubMed PMID: 6349785]
[50]
Cabrera AR, Kirkpatrick JP, Fiveash JB, Shih HA, Koay EJ, Lutz S, Petit J, Chao ST, Brown PD, Vogelbaum M, Reardon DA, Chakravarti A, Wen PY, Chang E. Radiation therapy for glioblastoma: Executive summary of an American Society for Radiation Oncology Evidence-Based Clinical Practice Guideline. Practical radiation oncology. 2016 Jul-Aug:6(4):217-225. doi: 10.1016/j.prro.2016.03.007. Epub 2016 Mar 31
[PubMed PMID: 27211230]
Level 1 (high-level) evidence
[51]
Sneed PK, Lamborn KR, Larson DA, Prados MD, Malec MK, McDermott MW, Weaver KA, Phillips TL, Wara WM, Gutin PH. Demonstration of brachytherapy boost dose-response relationships in glioblastoma multiforme. International journal of radiation oncology, biology, physics. 1996 Apr 1:35(1):37-44
[PubMed PMID: 8641924]
[52]
Koot RW, Maarouf M, Hulshof MC, Voges J, Treuer H, Koedooder C, Sturm V, Bosch DA. Brachytherapy: Results of two different therapy strategies for patients with primary glioblastoma multiforme. Cancer. 2000 Jun 15:88(12):2796-802
[PubMed PMID: 10870063]
[53]
Hunt CM, Thomas V, Alexander J. Glioblastoma Multiforme Survivor With Radiation-Induced Consequences: A Case Report. Cureus. 2022 Sep:14(9):e29397. doi: 10.7759/cureus.29397. Epub 2022 Sep 21
[PubMed PMID: 36304371]
Level 3 (low-level) evidence
[54]
Hegi ME, Diserens AC, Gorlia T, Hamou MF, de Tribolet N, Weller M, Kros JM, Hainfellner JA, Mason W, Mariani L, Bromberg JE, Hau P, Mirimanoff RO, Cairncross JG, Janzer RC, Stupp R. MGMT gene silencing and benefit from temozolomide in glioblastoma. The New England journal of medicine. 2005 Mar 10:352(10):997-1003
[PubMed PMID: 15758010]
[55]
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups, National Cancer Institute of Canada Clinical Trials Group. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. The New England journal of medicine. 2005 Mar 10:352(10):987-96
[PubMed PMID: 15758009]
[56]
Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, Ludwin SK, Allgeier A, Fisher B, Belanger K, Hau P, Brandes AA, Gijtenbeek J, Marosi C, Vecht CJ, Mokhtari K, Wesseling P, Villa S, Eisenhauer E, Gorlia T, Weller M, Lacombe D, Cairncross JG, Mirimanoff RO, European Organisation for Research and Treatment of Cancer Brain Tumour and Radiation Oncology Groups, National Cancer Institute of Canada Clinical Trials Group. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. The Lancet. Oncology. 2009 May:10(5):459-66. doi: 10.1016/S1470-2045(09)70025-7. Epub 2009 Mar 9
[PubMed PMID: 19269895]
Level 1 (high-level) evidence
[57]
Taphoorn MJ, Stupp R, Coens C, Osoba D, Kortmann R, van den Bent MJ, Mason W, Mirimanoff RO, Baumert BG, Eisenhauer E, Forsyth P, Bottomley A, European Organisation for Research and Treatment of Cancer Brain Tumour Group, EORTC Radiotherapy Group, National Cancer Institute of Canada Clinical Trials Group. Health-related quality of life in patients with glioblastoma: a randomised controlled trial. The Lancet. Oncology. 2005 Dec:6(12):937-44
[PubMed PMID: 16321761]
Level 2 (mid-level) evidence
[58]
Herrlinger U, Tzaridis T, Mack F, Steinbach JP, Schlegel U, Sabel M, Hau P, Kortmann RD, Krex D, Grauer O, Goldbrunner R, Schnell O, Bähr O, Uhl M, Seidel C, Tabatabai G, Kowalski T, Ringel F, Schmidt-Graf F, Suchorska B, Brehmer S, Weyerbrock A, Renovanz M, Bullinger L, Galldiks N, Vajkoczy P, Misch M, Vatter H, Stuplich M, Schäfer N, Kebir S, Weller J, Schaub C, Stummer W, Tonn JC, Simon M, Keil VC, Nelles M, Urbach H, Coenen M, Wick W, Weller M, Fimmers R, Schmid M, Hattingen E, Pietsch T, Coch C, Glas M, Neurooncology Working Group of the German Cancer Society. Lomustine-temozolomide combination therapy versus standard temozolomide therapy in patients with newly diagnosed glioblastoma with methylated MGMT promoter (CeTeG/NOA-09): a randomised, open-label, phase 3 trial. Lancet (London, England). 2019 Feb 16:393(10172):678-688. doi: 10.1016/S0140-6736(18)31791-4. Epub 2019 Feb 14
[PubMed PMID: 30782343]
Level 1 (high-level) evidence
[59]
Martínez-Garcia M, Álvarez-Linera J, Carrato C, Ley L, Luque R, Maldonado X, Martínez-Aguillo M, Navarro LM, Vaz-Salgado MA, Gil-Gil M. SEOM clinical guidelines for diagnosis and treatment of glioblastoma (2017). Clinical & translational oncology : official publication of the Federation of Spanish Oncology Societies and of the National Cancer Institute of Mexico. 2018 Jan:20(1):22-28. doi: 10.1007/s12094-017-1763-6. Epub 2017 Oct 30
[PubMed PMID: 29086250]
[60]
Zarnett OJ, Sahgal A, Gosio J, Perry J, Berger MS, Chang S, Das S. Treatment of elderly patients with glioblastoma: a systematic evidence-based analysis. JAMA neurology. 2015 May:72(5):589-96. doi: 10.1001/jamaneurol.2014.3739. Epub
[PubMed PMID: 25822375]
Level 1 (high-level) evidence
[61]
Herrlinger U, Schäfer N, Steinbach JP, Weyerbrock A, Hau P, Goldbrunner R, Friedrich F, Rohde V, Ringel F, Schlegel U, Sabel M, Ronellenfitsch MW, Uhl M, Maciaczyk J, Grau S, Schnell O, Hänel M, Krex D, Vajkoczy P, Gerlach R, Kortmann RD, Mehdorn M, Tüttenberg J, Mayer-Steinacker R, Fietkau R, Brehmer S, Mack F, Stuplich M, Kebir S, Kohnen R, Dunkl E, Leutgeb B, Proescholdt M, Pietsch T, Urbach H, Belka C, Stummer W, Glas M. Bevacizumab Plus Irinotecan Versus Temozolomide in Newly Diagnosed O6-Methylguanine-DNA Methyltransferase Nonmethylated Glioblastoma: The Randomized GLARIUS Trial. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2016 May 10:34(14):1611-9. doi: 10.1200/JCO.2015.63.4691. Epub 2016 Mar 14
[PubMed PMID: 26976423]
Level 1 (high-level) evidence
[62]
Rao AM, Quddusi A, Shamim MS. The significance of MGMT methylation in Glioblastoma Multiforme prognosis. JPMA. The Journal of the Pakistan Medical Association. 2018 Jul:68(7):1137-1139
[PubMed PMID: 30317322]
[63]
Pour ME, Moghadam SG, Shirkhani P, Sahebkar A, Mosaffa F. Therapeutic cell-based vaccines for glioblastoma multiforme. Medical oncology (Northwood, London, England). 2023 Nov 12:40(12):354. doi: 10.1007/s12032-023-02220-5. Epub 2023 Nov 12
[PubMed PMID: 37952224]
[64]
Iyer K, Saini S, Bhadra S, Kulavi S, Bandyopadhyay J. Precision medicine advancements in glioblastoma: A systematic review. BioMedicine. 2023:13(2):1-13. doi: 10.37796/2211-8039.1403. Epub 2023 Jun 1
[PubMed PMID: 37937301]
Level 1 (high-level) evidence
[65]
Gu Z, Wang Q, Chen J, Zhu Y. Predicted factors of surgical site infection in glioblastoma patients: A meta-analysis. International wound journal. 2023 Dec 3:21(3):e14504. doi: 10.1111/iwj.14504. Epub 2023 Dec 3
[PubMed PMID: 38044279]
Level 1 (high-level) evidence
[66]
Trevisi G, Mangiola A. Current Knowledge about the Peritumoral Microenvironment in Glioblastoma. Cancers. 2023 Nov 17:15(22):. doi: 10.3390/cancers15225460. Epub 2023 Nov 17
[PubMed PMID: 38001721]
[67]
Davis ME. Glioblastoma: Overview of Disease and Treatment. Clinical journal of oncology nursing. 2016 Oct 1:20(5 Suppl):S2-8. doi: 10.1188/16.CJON.S1.2-8. Epub
[PubMed PMID: 27668386]
Level 3 (low-level) evidence