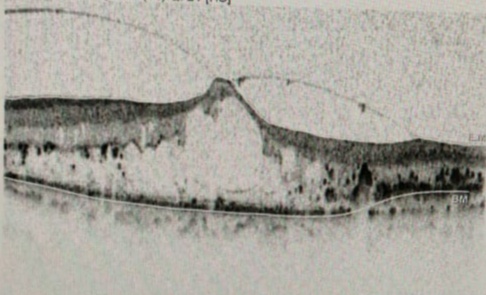
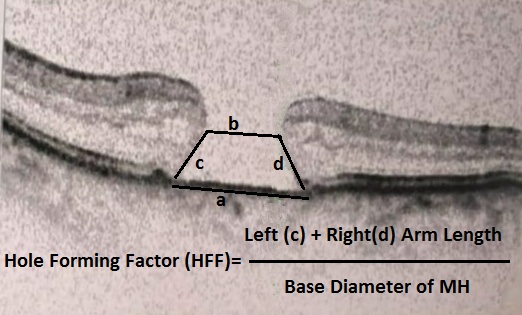
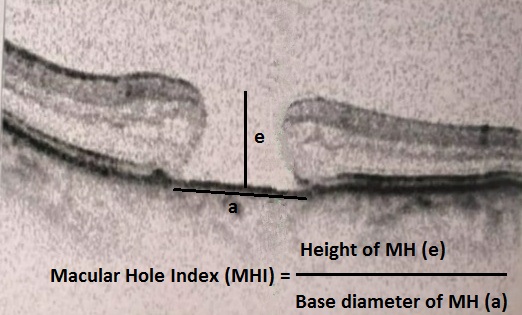
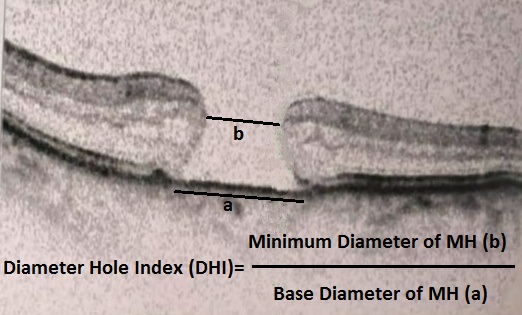
[1]
Gattoussi S, Buitendijk GHS, Peto T, Leung I, Schmitz-Valckenberg S, Oishi A, Wolf S, Deák G, Delcourt C, Klaver CCW, Korobelnik JF, European Eye Epidemiology (E3) consortium. The European Eye Epidemiology spectral-domain optical coherence tomography classification of macular diseases for epidemiological studies. Acta ophthalmologica. 2019 Jun:97(4):364-371. doi: 10.1111/aos.13883. Epub 2018 Sep 22
[PubMed PMID: 30242982]
Level 2 (mid-level) evidence
[2]
Azzolini C. Macular Hole: From Diagnosis to Therapy. Journal of ophthalmology. 2020:2020():1473763. doi: 10.1155/2020/1473763. Epub 2020 Mar 14
[PubMed PMID: 32280514]
[3]
Stein GE, Jung JJ, Bodine S, Trokel SL, Chang S. Vitrectomy for macular hole following Nd:YAG laser injury. Taiwan journal of ophthalmology. 2016 Oct-Dec:6(4):195-198. doi: 10.1016/j.tjo.2016.05.005. Epub 2016 Jun 20
[PubMed PMID: 29018741]
[4]
Pillai GS, Varkey R, Unnikrishnan UG, Radhakrishnan N. Incidence and risk factors for intraocular pressure rise after transconjunctival vitrectomy. Indian journal of ophthalmology. 2020 May:68(5):812-817. doi: 10.4103/ijo.IJO_244_19. Epub
[PubMed PMID: 32317451]
[5]
Chino M, Yoshikawa Y, Kanno J, Nagashima T, Sakaki Y, Katsumoto T, Shibuya M, Shoji T, Makita J, Shinoda K. Development and spontaneous closure of a secondary macular hole associated with submacular hemorrhage due to polypoidal choroidal vasculopathy: a case report. BMC ophthalmology. 2020 Mar 17:20(1):108. doi: 10.1186/s12886-020-01370-8. Epub 2020 Mar 17
[PubMed PMID: 32183733]
Level 3 (low-level) evidence
[6]
Peart S, Ramsay A, Khan QA, Leong T, Gordon-Bennett P. Large, Spontaneous Macular Hole with Posterior Pole Detachment in a Patient with Best Vitelliform Macular Dystrophy. Case reports in ophthalmology. 2019 May-Aug:10(2):221-226. doi: 10.1159/000501845. Epub 2019 Aug 6
[PubMed PMID: 31692623]
Level 3 (low-level) evidence
[7]
Tsuiki E, Kusano M, Kitaoka T. Complication associated with intravitreal injection of tissue plasminogen activator for treatment of submacular hemorrhage due to rupture of retinal arterial macroaneurysm. American journal of ophthalmology case reports. 2019 Dec:16():100556. doi: 10.1016/j.ajoc.2019.100556. Epub 2019 Sep 19
[PubMed PMID: 31650084]
Level 3 (low-level) evidence
[8]
Levison AL, Schachat AP. Macular hole from a central retinal artery occlusion. JAMA ophthalmology. 2014 Dec:132(12):1493. doi: 10.1001/jamaophthalmol.2014.549. Epub
[PubMed PMID: 25275337]
[9]
Fragiotta S, Rossi T, Carnevale C, Cutini A, Tricarico S, Casillo L, Scuderi G, Vingolo EM. Vitreo-macular interface disorders in retinitis pigmentosa. Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. 2019 Oct:257(10):2137-2146. doi: 10.1007/s00417-019-04418-8. Epub 2019 Jul 19
[PubMed PMID: 31324966]
[10]
Raimundo M, Fonseca C, Silva R, Figueira J. Bilateral giant macular holes: A rare manifestation of Alport syndrome. European journal of ophthalmology. 2019 Jan:29(1):NP13-NP16. doi: 10.1177/1120672118781232. Epub 2018 Jun 6
[PubMed PMID: 29873249]
[11]
Xie ZG, Yu SQ, Chen X, Zhu J, Chen F. Macular hole secondary to Valsalva retinopathy after doing push-up exercise. BMC ophthalmology. 2014 Aug 12:14():98. doi: 10.1186/1471-2415-14-98. Epub 2014 Aug 12
[PubMed PMID: 25117955]
[12]
Rizzo S, Mucciolo DP, Bacherini D, Murro V, Vannozzi L, Virgili G, Bani D, Sodi A. Macular hole in Stargardt disease: Clinical and ultra-structural observation. Ophthalmic genetics. 2017 Sep-Oct:38(5):486-489. doi: 10.1080/13816810.2016.1266666. Epub 2017 Jan 25
[PubMed PMID: 28121212]
[13]
Li Y, Li Z, Xu C, Liu Y, Kang X, Wu J. Autologous neurosensory retinal transplantation for recurrent macular hole retinal detachment in highly myopic eyes. Acta ophthalmologica. 2020 Dec:98(8):e983-e990. doi: 10.1111/aos.14442. Epub 2020 Apr 23
[PubMed PMID: 32323479]
[14]
Liao DY, Liu JH, Zheng YP, Shiu HW, Wang JM, Chao HM. OCT proves that vitreomacular adhesion is significantly more likely to develop vision-threatening retinal complications than vitreomacular separation. BMC ophthalmology. 2020 Apr 22:20(1):163. doi: 10.1186/s12886-020-01416-x. Epub 2020 Apr 22
[PubMed PMID: 32321473]
[15]
Gao M, Liu K, Lin Q, Liu H. Management Modalities for Traumatic Macular Hole: A Systematic Review and Single-Arm Meta-Analysis. Current eye research. 2017 Feb:42(2):287-296. doi: 10.1080/02713683.2016.1175021. Epub 2016 Jul 15
[PubMed PMID: 27420902]
Level 1 (high-level) evidence
[16]
Chen HJ, Jin Y, Shen LJ, Wang Y, Li ZY, Fang XY, Wang ZL, Huang XD, Wang ZJ, Ma ZZ. Traumatic macular hole study: a multicenter comparative study between immediate vitrectomy and six-month observation for spontaneous closure. Annals of translational medicine. 2019 Dec:7(23):726. doi: 10.21037/atm.2019.12.20. Epub
[PubMed PMID: 32042742]
Level 2 (mid-level) evidence
[17]
Lewis ML, Cohen SM, Smiddy WE, Gass JD. Bilaterality of idiopathic macular holes. Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. 1996 Apr:234(4):241-5
[PubMed PMID: 8964529]
[18]
Trempe CL, Weiter JJ, Furukawa H. Fellow eyes in cases of macular hole. Biomicroscopic study of the vitreous. Archives of ophthalmology (Chicago, Ill. : 1960). 1986 Jan:104(1):93-5
[PubMed PMID: 3942551]
Level 3 (low-level) evidence
[19]
la Cour M, Friis J. Macular holes: classification, epidemiology, natural history and treatment. Acta ophthalmologica Scandinavica. 2002 Dec:80(6):579-87
[PubMed PMID: 12485276]
[20]
McCannel CA, Ensminger JL, Diehl NN, Hodge DN. Population-based incidence of macular holes. Ophthalmology. 2009 Jul:116(7):1366-9. doi: 10.1016/j.ophtha.2009.01.052. Epub
[PubMed PMID: 19576500]
[21]
Ali FS, Stein JD, Blachley TS, Ackley S, Stewart JM. Incidence of and Risk Factors for Developing Idiopathic Macular Hole Among a Diverse Group of Patients Throughout the United States. JAMA ophthalmology. 2017 Apr 1:135(4):299-305. doi: 10.1001/jamaophthalmol.2016.5870. Epub
[PubMed PMID: 28208188]
[22]
. Risk factors for idiopathic macular holes. The Eye Disease Case-Control Study Group. American journal of ophthalmology. 1994 Dec 15:118(6):754-61
[PubMed PMID: 7977602]
Level 2 (mid-level) evidence
[23]
Morgan CM, Schatz H. Involutional macular thinning. A pre-macular hole condition. Ophthalmology. 1986 Feb:93(2):153-61
[PubMed PMID: 3951821]
[24]
Ozawa Y, Shinoda H, Nagai N, Tsubota K. Dynamic changes in neural retinal images during the development of a lamellar macular hole: A case report. Medicine. 2019 Dec:98(49):e18297. doi: 10.1097/MD.0000000000018297. Epub
[PubMed PMID: 31804376]
Level 3 (low-level) evidence
[25]
Green WR. The macular hole: histopathologic studies. Archives of ophthalmology (Chicago, Ill. : 1960). 2006 Mar:124(3):317-21
[PubMed PMID: 16534050]
[26]
Liu J, Peng J, Zhang Q, Ma M, Zhang H, Zhao P. Etiologies, Characteristics, and Management of Pediatric Macular Hole. American journal of ophthalmology. 2020 Feb:210():174-183. doi: 10.1016/j.ajo.2019.09.014. Epub 2019 Sep 24
[PubMed PMID: 31560879]
[27]
Xin W, Cai X, Xiao Y, Ji L, Gu Y, Lv W, Jiang J. Surgical treatment for type II macular hole retinal detachment in pathologic myopia. Medicine. 2020 Apr:99(17):e19531. doi: 10.1097/MD.0000000000019531. Epub
[PubMed PMID: 32332602]
[28]
Ezra E, Munro PM, Charteris DG, Aylward WG, Luthert PJ, Gregor ZJ. Macular hole opercula. Ultrastructural features and clinicopathological correlation. Archives of ophthalmology (Chicago, Ill. : 1960). 1997 Nov:115(11):1381-7
[PubMed PMID: 9366667]
[29]
Sebag J. [The vitreoretinal interface and its role in the pathogenesis of vitreomaculopathies]. Der Ophthalmologe : Zeitschrift der Deutschen Ophthalmologischen Gesellschaft. 2015 Jan:112(1):10-9. doi: 10.1007/s00347-014-3048-6. Epub
[PubMed PMID: 25681055]
[30]
Quinn NB, Steel DH, Chakravarthy U, Peto T, Hamill B, Muldrew A, Graham K, Elliott D, Hennessy R, Cruise S, McGuinness B, Young IS, Kee F, Hogg RE. Assessment of the Vitreomacular Interface Using High-Resolution OCT in a Population-Based Cohort Study of Older Adults. Ophthalmology. Retina. 2020 Aug:4(8):801-813. doi: 10.1016/j.oret.2020.02.013. Epub 2020 Feb 29
[PubMed PMID: 32335034]
[31]
Folk JC, Boldt HC, Keenum DG. Foveal cysts: a premacular hole condition associated with vitreous traction. Archives of ophthalmology (Chicago, Ill. : 1960). 1998 Sep:116(9):1177-83
[PubMed PMID: 9747675]
[32]
Cicinelli MV, Marchese A, Bandello F, Coppola M. Inner Retinal Layer and Outer Retinal Layer Findings after Macular Hole Surgery Assessed by means of Optical Coherence Tomography. Journal of ophthalmology. 2019:2019():3821479. doi: 10.1155/2019/3821479. Epub 2019 Apr 1
[PubMed PMID: 31061725]
[33]
Saxena S, Holekamp NM, Kumar A. Diagnosis and management of idiopathic macular holes. Indian journal of ophthalmology. 1998 Dec:46(4):185-93
[PubMed PMID: 10218300]
[34]
Venkatesh R, Mohan A, Sinha S, Aseem A, Yadav NK. Newer indices for predicting macular hole closure in idiopathic macular holes: A retrospective, comparative study. Indian journal of ophthalmology. 2019 Nov:67(11):1857-1862. doi: 10.4103/ijo.IJO_364_19. Epub
[PubMed PMID: 31638049]
Level 2 (mid-level) evidence
[35]
Sjaarda RN, Frank DA, Glaser BM, Thompson JT, Murphy RP. Resolution of an absolute scotoma and improvement of relative scotomata after successful macular hole surgery. American journal of ophthalmology. 1993 Aug 15:116(2):129-39
[PubMed PMID: 8352296]
[37]
Kelly NE, Wendel RT. Vitreous surgery for idiopathic macular holes. Results of a pilot study. Archives of ophthalmology (Chicago, Ill. : 1960). 1991 May:109(5):654-9
[PubMed PMID: 2025167]
Level 3 (low-level) evidence
[38]
Liang X, Liu W. Characteristics and Risk Factors for Spontaneous Closure of Idiopathic Full-Thickness Macular Hole. Journal of ophthalmology. 2019:2019():4793764. doi: 10.1155/2019/4793764. Epub 2019 Mar 13
[PubMed PMID: 31001430]
[39]
Khan MA, Haller JA. Ocriplasmin for Treatment of Vitreomacular Traction: An Update. Ophthalmology and therapy. 2016 Dec:5(2):147-159
[PubMed PMID: 27619226]
[40]
Eckardt C, Eckardt U, Groos S, Luciano L, Reale E. [Removal of the internal limiting membrane in macular holes. Clinical and morphological findings]. Der Ophthalmologe : Zeitschrift der Deutschen Ophthalmologischen Gesellschaft. 1997 Aug:94(8):545-51
[PubMed PMID: 9376691]
[41]
Muqit MMK, Hamilton R, Ho J, Tucker S, Buck H. Intravitreal ocriplasmin for the treatment of vitreomacular traction and macular hole- A study of efficacy and safety based on NICE guidance. PloS one. 2018:13(5):e0197072. doi: 10.1371/journal.pone.0197072. Epub 2018 May 16
[PubMed PMID: 29768451]
Level 2 (mid-level) evidence
[42]
Juncal VR, Chow DR, Vilà N, Kapusta MA, Williams RG, Kherani A, Berger AR. Ocriplasmin versus vitrectomy for the treatment of macular holes. Canadian journal of ophthalmology. Journal canadien d'ophtalmologie. 2018 Oct:53(5):441-446. doi: 10.1016/j.jcjo.2018.01.017. Epub 2018 Mar 21
[PubMed PMID: 30340707]
[43]
Prospero Ponce CM, Stevenson W, Gelman R, Agarwal DR, Christoforidis JB. Ocriplasmin: who is the best candidate? Clinical ophthalmology (Auckland, N.Z.). 2016:10():485-95. doi: 10.2147/OPTH.S97947. Epub 2016 Mar 17
[PubMed PMID: 27051270]
[44]
Rodrigues EB, Meyer CH. Meta-analysis of chromovitrectomy with indocyanine green in macular hole surgery. Ophthalmologica. Journal international d'ophtalmologie. International journal of ophthalmology. Zeitschrift fur Augenheilkunde. 2008:222(2):123-9. doi: 10.1159/000112630. Epub 2008 Feb 22
[PubMed PMID: 18303234]
Level 1 (high-level) evidence
[45]
Stalmans P, Van Aken EH, Veckeneer M, Feron EJ, Stalmans I. Toxic effect of indocyanine green on retinal pigment epithelium related to osmotic effects of the solvent. American journal of ophthalmology. 2002 Aug:134(2):282-5
[PubMed PMID: 12140045]
[46]
Shukla D, Kalliath J, Neelakantan N, Naresh KB, Ramasamy K. A comparison of brilliant blue G, trypan blue, and indocyanine green dyes to assist internal limiting membrane peeling during macular hole surgery. Retina (Philadelphia, Pa.). 2011 Nov:31(10):2021-5. doi: 10.1097/IAE.0b013e318213618c. Epub
[PubMed PMID: 21685824]
[47]
Michalewska Z, Michalewski J, Adelman RA, Nawrocki J. Inverted internal limiting membrane flap technique for large macular holes. Ophthalmology. 2010 Oct:117(10):2018-25. doi: 10.1016/j.ophtha.2010.02.011. Epub 2010 Jun 11
[PubMed PMID: 20541263]
[48]
Michalewska Z, Nawrocki J. Vitrectomy with the inverted internal limiting membrane flap technique in eyes with full-thickness macular hole and dry age-related macular degeneration. European journal of ophthalmology. 2021 May:31(3):1320-1325. doi: 10.1177/1120672120921376. Epub 2020 Apr 28
[PubMed PMID: 32345051]
[49]
Ye T, Yu JG, Liao L, Liu L, Xia T, Yang LL. Macular hole surgery recovery with and without face-down posturing: a meta-analysis of randomized controlled trials. BMC ophthalmology. 2019 Dec 21:19(1):265. doi: 10.1186/s12886-019-1272-1. Epub 2019 Dec 21
[PubMed PMID: 31864333]
Level 1 (high-level) evidence
[50]
Berger JW, Brucker AJ. The magnitude of the bubble buoyant pressure: implications for macular hole surgery. Retina (Philadelphia, Pa.). 1998:18(1):84-6; author reply 86-8
[PubMed PMID: 9502292]
[51]
Paques M, Massin P, Blain P, Duquesnoy AS, Gaudric A. Long-term incidence of reopening of macular holes. Ophthalmology. 2000 Apr:107(4):760-5; discussion 766
[PubMed PMID: 10768340]
[52]
Yamashita T, Sakamoto T, Yamashita T, Sonoda S, Yamakiri K, Otsuka H, Hisatomi T, Imaki H, Ishibashi T, Dugel PU. Individualized, spectral domain-optical coherence tomography-guided facedown posturing after macular hole surgery: minimizing treatment burden and maximizing outcome. Retina (Philadelphia, Pa.). 2014 Jul:34(7):1367-75. doi: 10.1097/IAE.0000000000000087. Epub
[PubMed PMID: 24955569]
[53]
Wirbelauer C, Kolarov D, Just A. [Influence of Macular Hole Width on Visual Acuity, Endotamponade and Closure Rate]. Klinische Monatsblatter fur Augenheilkunde. 2016 Dec:233(12):1362-1366
[PubMed PMID: 27984839]
[54]
Gass JD. Idiopathic senile macular hole: its early stages and pathogenesis. 1988. Retina (Philadelphia, Pa.). 2003 Dec:23(6 Suppl):629-39
[PubMed PMID: 15035400]
[55]
Duker JS, Kaiser PK, Binder S, de Smet MD, Gaudric A, Reichel E, Sadda SR, Sebag J, Spaide RF, Stalmans P. The International Vitreomacular Traction Study Group classification of vitreomacular adhesion, traction, and macular hole. Ophthalmology. 2013 Dec:120(12):2611-2619. doi: 10.1016/j.ophtha.2013.07.042. Epub 2013 Sep 17
[PubMed PMID: 24053995]
[56]
Ullrich S, Haritoglou C, Gass C, Schaumberger M, Ulbig MW, Kampik A. Macular hole size as a prognostic factor in macular hole surgery. The British journal of ophthalmology. 2002 Apr:86(4):390-3
[PubMed PMID: 11914205]
[57]
Kusuhara S, Teraoka Escaño MF, Fujii S, Nakanishi Y, Tamura Y, Nagai A, Yamamoto H, Tsukahara Y, Negi A. Prediction of postoperative visual outcome based on hole configuration by optical coherence tomography in eyes with idiopathic macular holes. American journal of ophthalmology. 2004 Nov:138(5):709-16
[PubMed PMID: 15531303]
[58]
Ruiz-Moreno JM, Staicu C, Piñero DP, Montero J, Lugo F, Amat P. Optical coherence tomography predictive factors for macular hole surgery outcome. The British journal of ophthalmology. 2008 May:92(5):640-4. doi: 10.1136/bjo.2007.136176. Epub
[PubMed PMID: 18441174]
[59]
Dai YM, Shen J, Li JK, Jin XH, Li YM. [Optical coherence tomography predictive factors for idiopathic macular hole surgery outcome]. [Zhonghua yan ke za zhi] Chinese journal of ophthalmology. 2013 Sep:49(9):807-11
[PubMed PMID: 24330930]
[60]
Shahlaee A, Rahimy E, Hsu J, Gupta OP, Ho AC. Preoperative and postoperative features of macular holes on en face imaging and optical coherence tomography angiography. American journal of ophthalmology case reports. 2017 Apr:5():20-25. doi: 10.1016/j.ajoc.2016.10.008. Epub 2016 Oct 29
[PubMed PMID: 29503940]
Level 3 (low-level) evidence
[61]
Chen GH, Tzekov R, Jiang FZ, Mao SH, Tong YH, Li WS. Iatrogenic retinal breaks and postoperative retinal detachments in microincision vitrectomy surgery compared with conventional 20-gauge vitrectomy: a meta-analysis. Eye (London, England). 2019 May:33(5):785-795. doi: 10.1038/s41433-018-0319-5. Epub 2018 Dec 18
[PubMed PMID: 30560911]
Level 1 (high-level) evidence
[62]
Li SS, You R, Li M, Guo XX, Zhao L, Wang YL, Chen X. Internal limiting membrane peeling with different dyes in the surgery of idiopathic macular hole: a systematic review of literature and network Meta-analysis. International journal of ophthalmology. 2019:12(12):1917-1928. doi: 10.18240/ijo.2019.12.15. Epub 2019 Dec 18
[PubMed PMID: 31850178]
Level 1 (high-level) evidence
[63]
Unsal E, Cubuk MO, Ciftci F. Preoperative prognostic factors for macular hole surgery: Which is better? Oman journal of ophthalmology. 2019 Jan-Apr:12(1):20-24. doi: 10.4103/ojo.OJO_247_2017. Epub
[PubMed PMID: 30787530]
[64]
Kang SW, Ahn K, Ham DI. Types of macular hole closure and their clinical implications. The British journal of ophthalmology. 2003 Aug:87(8):1015-9
[PubMed PMID: 12881347]
[65]
Park SS, Marcus DM, Duker JS, Pesavento RD, Topping TM, Frederick AR Jr, D'Amico DJ. Posterior segment complications after vitrectomy for macular hole. Ophthalmology. 1995 May:102(5):775-81
[PubMed PMID: 7777277]
[66]
Rizzo S, Belting C, Genovesi-Ebert F, di Bartolo E. Incidence of retinal detachment after small-incision, sutureless pars plana vitrectomy compared with conventional 20-gauge vitrectomy in macular hole and epiretinal membrane surgery. Retina (Philadelphia, Pa.). 2010 Jul-Aug:30(7):1065-71. doi: 10.1097/IAE.0b013e3181cd48b0. Epub
[PubMed PMID: 20616684]
[67]
Guillaubey A, Malvitte L, Lafontaine PO, Hubert I, Bron A, Berrod JP, Creuzot-Garcher C. Incidence of retinal detachment after macular surgery: a retrospective study of 634 cases. The British journal of ophthalmology. 2007 Oct:91(10):1327-30
[PubMed PMID: 17522152]
Level 2 (mid-level) evidence
[68]
Runkle AP, Srivastava SK, Yuan A, Kaiser PK, Singh RP, Reese JL, Ehlers JP. FACTORS ASSOCIATED WITH DEVELOPMENT OF DISSOCIATED OPTIC NERVE FIBER LAYER APPEARANCE IN THE PIONEER INTRAOPERATIVE OPTICAL COHERENCE TOMOGRAPHY STUDY. Retina (Philadelphia, Pa.). 2018 Sep:38 Suppl 1(Suppl 1):S103-S109. doi: 10.1097/IAE.0000000000002017. Epub
[PubMed PMID: 29346239]
[69]
Spaide RF. "Dissociated optic nerve fiber layer appearance" after internal limiting membrane removal is inner retinal dimpling. Retina (Philadelphia, Pa.). 2012 Oct:32(9):1719-26
[PubMed PMID: 23007669]
[70]
Tadayoni R, Svorenova I, Erginay A, Gaudric A, Massin P. Decreased retinal sensitivity after internal limiting membrane peeling for macular hole surgery. The British journal of ophthalmology. 2012 Dec:96(12):1513-6. doi: 10.1136/bjophthalmol-2012-302035. Epub 2012 Oct 17
[PubMed PMID: 23077227]
[71]
Paques M, Massin P, Santiago PY, Spielmann AC, Gaudric A. Visual field loss after vitrectomy for full-thickness macular holes. American journal of ophthalmology. 1997 Jul:124(1):88-94
[PubMed PMID: 9222237]
[72]
Ezra E, Arden GB, Riordan-Eva P, Aylward GW, Gregor ZJ. Visual field loss following vitrectomy for stage 2 and 3 macular holes. The British journal of ophthalmology. 1996 Jun:80(6):519-25
[PubMed PMID: 8759262]
[73]
Welch JC. Dehydration injury as a possible cause of visual field defect after pars plana vitrectomy for macular hole. American journal of ophthalmology. 1997 Nov:124(5):698-9
[PubMed PMID: 9372731]
[74]
Christmas NJ, Smiddy WE, Flynn HW Jr. Reopening of macular holes after initially successful repair. Ophthalmology. 1998 Oct:105(10):1835-8
[PubMed PMID: 9787352]
[75]
Rahimy E, McCannel CA. IMPACT OF INTERNAL LIMITING MEMBRANE PEELING ON MACULAR HOLE REOPENING: A Systematic Review and Meta-Analysis. Retina (Philadelphia, Pa.). 2016 Apr:36(4):679-87. doi: 10.1097/IAE.0000000000000782. Epub
[PubMed PMID: 26441264]
Level 1 (high-level) evidence
[76]
Paques M, Massin P, Santiago PY, Spielmann AC, Le Gargasson JF, Gaudric A. Late reopening of successfully treated macular holes. The British journal of ophthalmology. 1997 Aug:81(8):658-62
[PubMed PMID: 9349153]
[77]
Knight D, Yu JJ, Adrean SD. RELAXING NASAL RETINOTOMY TECHNIQUE FOR CLOSURE OF A MACULAR HOLE THAT REOPENED AFTER PRIMARY VITRECTOMY. Retinal cases & brief reports. 2021 Sep 1:15(5):611-614. doi: 10.1097/ICB.0000000000000862. Epub
[PubMed PMID: 30865057]
Level 3 (low-level) evidence
[78]
Rao X, Wang NK, Chen YP, Hwang YS, Chuang LH, Liu IC, Chen KJ, Wu WC, Lai CC. Outcomes of outpatient fluid-gas exchange for open macular hole after vitrectomy. American journal of ophthalmology. 2013 Aug:156(2):326-333.e1. doi: 10.1016/j.ajo.2013.03.031. Epub 2013 May 17
[PubMed PMID: 23688710]
[79]
Holekamp NM, Meredith TA, Landers MB, Snyder WB, Thompson JT, Berman AJ, Williams S. Ulnar neuropathy as a complication of macular hole surgery. Archives of ophthalmology (Chicago, Ill. : 1960). 1999 Dec:117(12):1607-10
[PubMed PMID: 10604664]
[80]
Fahim AT, Khan NW, Johnson MW. Acute panretinal structural and functional abnormalities after intravitreous ocriplasmin injection. JAMA ophthalmology. 2014 Apr 1:132(4):484-6. doi: 10.1001/jamaophthalmol.2013.8142. Epub
[PubMed PMID: 24577241]
[81]
Zandi S, Freiberg F, Vaclavik V, Pfister IB, Traine PG, Kaya C, Michels S, Garweg JG. Morphological Reconstitution and Persistent Changes After Intravitreal Ocriplasmin for Vitreomacular Traction and Macular Hole. Journal of ocular pharmacology and therapeutics : the official journal of the Association for Ocular Pharmacology and Therapeutics. 2020 Mar:36(2):126-132. doi: 10.1089/jop.2019.0051. Epub 2020 Jan 14
[PubMed PMID: 31934816]
[82]
Florent C, Coupier L, Mérité PY, Meyer F, Guigou S. Human amniotic membrane plug technique for macular hole surgery: A case report. Journal francais d'ophtalmologie. 2020 Apr:43(4):e151-e152. doi: 10.1016/j.jfo.2019.09.006. Epub 2020 Feb 25
[PubMed PMID: 32111503]
Level 3 (low-level) evidence
[83]
Caporossi T, Pacini B, De Angelis L, Barca F, Peiretti E, Rizzo S. HUMAN AMNIOTIC MEMBRANE TO CLOSE RECURRENT, HIGH MYOPIC MACULAR HOLES IN PATHOLOGIC MYOPIA WITH AXIAL LENGTH OF ≥30 mm. Retina (Philadelphia, Pa.). 2020 Oct:40(10):1946-1954. doi: 10.1097/IAE.0000000000002699. Epub
[PubMed PMID: 31868775]
[84]
Lai CC, Chen YP, Wang NK, Chuang LH, Liu L, Chen KJ, Hwang YS, Wu WC, Chen TL. Vitrectomy with Internal Limiting Membrane Repositioning and Autologous Blood for Macular Hole Retinal Detachment in Highly Myopic Eyes. Ophthalmology. 2015 Sep:122(9):1889-98. doi: 10.1016/j.ophtha.2015.05.040. Epub 2015 Jul 2
[PubMed PMID: 26143541]
[85]
Kapoor KG, Khan AN, Tieu BC, Khurshid GS. Revisiting autologous platelets as an adjuvant in macular hole repair: chronic macular holes without prone positioning. Ophthalmic surgery, lasers & imaging : the official journal of the International Society for Imaging in the Eye. 2012 Jul 1:43(4):291-5. doi: 10.3928/15428877-20120426-03. Epub 2012 May 3
[PubMed PMID: 22589336]
[86]
Zhang X, Liu J, Yu B, Ma F, Ren X, Li X. Effects of mesenchymal stem cells and their exosomes on the healing of large and refractory macular holes. Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. 2018 Nov:256(11):2041-2052. doi: 10.1007/s00417-018-4097-3. Epub 2018 Aug 30
[PubMed PMID: 30167916]