 Nuclear Medicine PET/CT Gastrointestinal Assessment, Protocols, and Interpretation
Nuclear Medicine PET/CT Gastrointestinal Assessment, Protocols, and Interpretation
Introduction
Gastrointestinal malignancies are a heterogeneous group of neoplasms encompassing diverse biological and physical behavior. The management options for these tumors vary based on their location, cell type, and growth pattern. Early precise diagnosis and accurate staging are critical to instituting the appropriate treatment. Recent advancements in imaging techniques for evaluating solid and hollow viscus organs have enabled the noninvasive assessment of these tumors, although histopathology remains the gold standard. Despite these refinements, no single imaging modality provides sufficient information, and often, multiple investigations are judicially combined to offer strength from one modality where the other is lacking.[1]
Positron emission tomography/computed tomography (PET/CT) is a hybrid molecular imaging technique that combines the functional imaging advantages of positron emission tomography (PET) with anatomical imaging from computed tomography (CT). The cancerous cells of the gastrointestinal tract (GIT) demonstrate preferential glucose metabolism to lactate in both aerobic and anaerobic environments, a phenomenon called the "Warburg effect."[2] The energy produced through this pathway is insufficient to meet the demand of increasing growth, raising the cells' glucose requirement. The oncogenes in the cell structures and growth factors activated by malignant cells trigger glycolysis through upregulation of glucose transporter 1 (GLUT-1) and cellular hexokinase, forming the biochemical basis of fluorodeoxyglucose-PET (FDG-PET) imaging.
Fluorine-18-labeled fluorodeoxyglucose (18F-FDG) is a glucose analog that accumulates in cancerous cells. However, unlike glucose, this labeled carbohydrate cannot be channeled toward adenosine triphosphate (ATP) production. This entrapment of FDG in cancerous and some inflammatory cells is responsible for image production in PET scanning. In contrast, gallium PET/CT imaging relies on the overexpression of somatostatin receptors (SSTRs) in well-differentiated neuroendocrine tumors (NETs), thus offering an exquisite assessment of the selected primary NETs and their metastases.[3]
Anatomy and Physiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Anatomy and Physiology
Esophagus and Gastroesophageal Junction
Optimum treatment planning of esophageal cancers requires an accurate description of cancer location in upper gastrointestinal tumors, which is impossible without a clear understanding of the segmental esophageal anatomy. Anatomical levels for these cancers are reported relative to anatomical landmarks set by the American Joint Committee on Cancer (AJCC) staging system.
The esophagus is divided into 4 segments. This delineation is vital to ensure that the tumor is covered in its entirety in the radiotherapy field. If surgical resection is planned, the choice of approach is appropriate for the tumor's location. The Siewert-Stein classification categorizes gastroesophageal junction (GEJ) tumors into 3 groups based on their epicenter's location relative to the gastroesophageal junction. Regional lymph nodes for the esophagus include the supraclavicular, periesophageal, and celiac nodes.[4]
Large Intestine
The large intestine extends from the cecum to the anal verge, where the stratified squamous epithelium of the anal canal meets the anal skin. For colorectal cancers, the tumor's relationship with the peritoneal reflection carries significant prognostic value. Peritoneal involvement classifies the disease as T4a and increases the likelihood of peritoneal metastases.[5]
The parietal peritoneum covering the abdominal wall reflects anteriorly onto the cecum and the ascending and descending colon, becoming the visceral peritoneum while leaving their posterior circumference uncovered. The transverse and sigmoid colon are almost entirely intraperitoneal, except for the areas along the mesenteric vessels. The peritoneal reflection on the pelvic structures forms the rectouterine pouch in the female body and the rectovesical pouch in the male body, partially covering the pelvic sidewall on both sides. The rectum is covered by the peritoneum anteriorly and laterally in the upper part, only anteriorly in the middle part, and is entirely free of the peritoneum in the lower part.
The anal canal is the terminal segment of the GIT and extends from the anorectal junction superiorly, where the rectum passes through the pelvic diaphragm to the anal verge inferiorly. This segment measures approximately 4 cm and has 2 crucial muscle layers. The inner layer, which is the continuation of the GIT's circular muscle, forms the internal sphincter. The outer layer, derived from the downward continuation of the levator ani and puborectalis, forms the external sphincter, with a small intersphincteric space separating the internal and external sphincters.
Nodal metastases from colon cancer spread along the mesenteric vessels to the origin of the superior mesenteric artery (SMA) in right-sided tumors or the inferior mesenteric artery (IMA) in left-sided tumors, potentially involving the paraaortic nodes. Rectal tumors drain into the mesorectal nodes, then to the superior rectal artery nodes. From there, metastases can travel along the sigmoid mesentery root to the paraaortic nodes or drain into the internal iliac nodes.
Anal cancer has a greater predilection for spreading through the lymphatic system rather than the bloodstream.[6] Tumors superior to the dentate line follow the drainage pathway of rectal tumors, while those inferior to the dentate line have a predilection for draining into the external iliac system via the inguinal and femoral nodes.
Neuroendocrine Tumors
Gastroenteropancreatic NETs (GEP-NETs) are a diverse group of neoplasms originating from embryonal neural crest cells. These tumors can develop in virtually any part of the GIT, with the small intestine being the most common location. Gastroenteropancreatic NETs exhibit a wide range of clinical behaviors, from indolent, slow-growing forms to highly aggressive variants. This variability in clinical behavior correlates with the tumors' cellular proliferation rate, which is measured using immunohistochemical staining for the Ki67 protein.
According to the European Neuroendocrine Tumour Society and World Health Organization 2010 Classification Systems for NETs, these tumors are categorized into 3 grades based on the Ki67 index. Grade 1 tumors are well-differentiated with a Ki67 index below 2% and demonstrate indolent behavior. Grade 3 tumors are poorly differentiated, with a Ki67 index above 20%, and exhibit aggressive clinical behavior. Grade 2 tumors, which are moderately differentiated, display a behavior and proliferation profile between these 2 extremes.[7][8]
Radiotracers for Positron Emission Tomography/Computed Tomography Imaging
The most commonly used radiotracer in PET/CT imaging is 18F-FDG. Activity detection occurs through multiple collisions between the positron emitted from the radioactive material and surrounding electrons in the biological environment. This process continues until the positron loses its kinetic energy and combines with an electron to form a short-lived positronium molecule. The positronium then undergoes annihilation, converting its mass into 2 γ-ray photons, each having an energy of 511 keV and traveling in opposite directions 180° apart.
A coincidence event occurs when both annihilation photons are detected within a short time interval, and the photons are assumed to have originated from positronium annihilation along the line connecting the 2 points. This process eliminates the need for a physical lead collimator, resulting in significantly better sensitivity compared to single photon emission computed tomography (SPECT) imaging, as no extrinsic collimation is required.[9]
In gallium imaging, the conjugated version of octreotide is attached to a gallium-68 radiotracer through a 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA) ligand. This conjugation allows the radiotracer to effectively bind with SSTRs found on well-differentiated NETs. However, more aggressive, poorly differentiated tumors may not show significant uptake on gallium PET and are sometimes better assessed with FDG-PET due to their lower expression of SSTRs and increased glucose metabolism.[10]
Indications
Evidence-based indications for the use of FDG-PET/CT for gastrointestinal tumors in the United Kingdom, as published by the UK Royal College of Radiologists in 2016, include the following:
- For esophageal and gastroesophageal cancers, PET/CT is indicated for staging and restaging when radical treatment is appropriate, including in patients who have received neoadjuvant therapy. PET/CT is also useful for evaluating suspected recurrence when other imaging techniques yield negative or equivocal results. Additional proposed indications include response assessment and radiotherapy planning.
- For colorectal cancers, PET/CT is indicated for staging disease with synchronous metastases at presentation, such as pulmonary and liver lesions. Other indications include restaging in patients with recurrence, assessing treatment response, detecting recurrence in the presence of rising tumor markers, and evaluating clinical suspicion of recurrence when other imaging modalities have normal or equivocal results. Posttreatment, PET/CT may also be used to evaluate indeterminate presacral masses.
- PET/CT is performed in anal cancer for staging in selected patients requiring radical treatment. Additional proposed indications include radiotherapy planning and response assessment.
- For NETs, FDG-PET/CT is indicated for staging and restaging poorly differentiated tumors that show normal or negative m-iodobenzylguanidine (MIBG) and octreotide scans. Gallium-68 DOTA-[1-Naphthylmethyl] octreotide PET/CT (68Ga-DOTANOC PET/CT) is indicated for staging well-differentiated NETs (grades 1 and 2), detecting unknown primaries, and evaluating recurrence (see Image. Gastrointestinal Neuroendocrine Tumor on Advanced Nuclear Imaging).
Contraindications
Imaging with PET/CT requires careful patient preparation. Poor glycemic control, incomplete fasting, and insulin injection immediately before the scan can affect the radiotracer's biodistribution. Elevated blood glucose competes with FDG for binding to GLUT-1 transporters, while high insulin levels can redirect a significant amount of FDG to skeletal muscles and adipose tissue via insulin-dependent glucose transporter type 4 (GLUT-4).
Ideally, PET/CT should be delayed for at least 6 weeks after radiotherapy or surgery and at least 2 weeks after stent placement to prevent inflammatory uptake at the intervention site, which may lead to overestimation of the disease.[11]
Patients with claustrophobia may require sedation. PET/CT is generally unsuitable for pregnant patients, and an alternative means of evaluating disease in this group should be arranged. Breastfeeding mothers should avoid contact with their infants after the scan. The general recommendation is to have the infants bottle-fed by a 3rd person for up to 12 hours after the scan.[12][13]
Equipment
The radiopharmaceutical is prepared in cyclotrons and brought into the department in lead-lined containers early in the morning. The handling process requires exquisite care and diligence to avoid exposure to the staff and the public. The radiotracer is injected through a tungsten syringe employed in a specialized carrier and is kept behind a lead screen.[14]
The PET/CT system comprises stand-alone PET and CT scanners housed in a single gantry. The patient bed is shared between the 2 scanners and moves smoothly from the PET to the CT component during image acquisition.
A scintillator absorbs ionizing radiation and converts a fraction of the absorbed energy into photons of visible or ultraviolet light, which are then transformed into electrical signals by photodetectors. A typical PET scanner consists of thousands of inorganic scintillation crystals organized into blocks, which form detector modules arranged in a ring configuration. Traditionally, bismuth germanate (BGO) detectors were used, but they have now been replaced by lutetium oxyorthosilicate (LSO) detectors.[15]
These crystals absorb energy from annihilation events in the body (511 keV energy) and generate tens of thousands of visible wavelength photons, proportional to the energy deposited by the annihilation photons. "Random coincidence" occurs when 2 detected photons originate from separate annihilation events. "Scatter coincidence" happens when both photons are from the same event, but 1 or both have scattered. "True coincidence" refers to the detection of both photons from the same annihilation event.[16]
The light photons are channeled toward the coupled photodetectors. The most common photodetectors used in PET are photomultiplier tubes (PMTs), which consist of vacuum tubes with photocathodes. The light photons interact with the cathode, producing electrons that are then amplified to generate an electric signal proportional to the energy deposited by the PET photons.
The initial image is far from perfect and is significantly influenced by PET photon attenuation in the body, variations in detector efficiency, and the recording of scattering and random events alongside true coincidences. Consequently, attenuation correction is performed using a CT scan conducted simultaneously. The CT component also provides additional anatomical information that is absent in the initial PET images.
The images are then represented quantitatively in terms of radioactivity concentration (in kBq/ml), which is converted into a standardized uptake value (SUV) by normalizing to the patient's weight and the injected radioactivity.[17] This SUV measurement quantifies glucose metabolism in a specific tissue of the body and is calculated as follows:
SUV (g/ml) = [(tracer uptake in kBq/ml) / (activity in MBq / patient's weight in kg)]
Preparation
Patient preparation for FDG-PET is complex because FDG, a glucose analog, is absorbed by tissues that normally transport glucose. As PET imaging assesses cellular-level function, it is susceptible to artifacts caused by physiological increases in cellular activity. Managing glycemic levels and minimizing activity in nontarget areas are crucial to maximizing diagnostic accuracy.
Glycemic Control
Patients must fast for 4 to 6 hours prior to the scan to reduce competition between glucose and deoxyglucose at GLUT-1 transporters. Eating induces an insulin spike, which can divert glucose/FDG to peripheral muscles via insulin-dependent GLUT-4 receptors. In patients with diabetes mellitus, imaging is best performed in the early morning, utilizing the benefits of overnight fasting. For these reasons, insulin should not be injected before the scan. Blood glucose levels are checked beforehand to ensure they are below 200 mg/dl.[18]
Muscular Activity
Patients should refrain from exercise the day before and the day of the procedure. During the scan, patients must remain relaxed and avoid swallowing or speaking to prevent radiotracer uptake in the muscles involved in phonation.
Brown Fat Uptake
Brown fat, which produces nonshivering thermogenesis, can take up FDG in cold environments, potentially leading to false-positive PET results. A warm environment must be maintained for the patient to control brown fat activity.
Hydration
Patients should be well-hydrated to promote renal excretion of FDG. Patients are also instructed to empty their bladder just before the scan to reduce activity in the pelvic area caused by a full bladder. After the scan, patients are advised to drink water to help facilitate the excretion of radioactive material from the body. Caffeinated drinks are not recommended.
Cannulation
The procedure is thoroughly explained to patients, and any questions are addressed to minimize anxiety. Patients are then seated in a lead-lined room, where cannulation should be performed to minimize radiation exposure to staff members. A butterfly infusion set is used.
The patient's vaccine status and injection sites are documented to prevent false-positive results. Local inflammation at the injection site can cause FDG-avid reactive changes in the draining lymph nodes, commonly observed in the axilla following arm vaccinations.[19]
Technique or Treatment
The recommended intravenous dose of 18F-FDG for adults ranges from 370 to 740 MBq, while for children, it is 4 to 5 MBq/kg.[20] After injection, patients are seated comfortably in a warm environment for 1 hour to allow proper FDG distribution. Any radiotracer extravasation is documented, and patients are instructed to empty their bladder just before scanning.
Imaging is typically conducted without the use of oral or intravenous contrast. Patients are routinely encouraged to drink water, which can serve as a negative contrast agent for the GIT.
Once the patient is positioned on the scanner bed, image acquisition begins with a topogram to plan the scanning area. For gastrointestinal cancers, imaging is typically performed from the base of the skull to the midthigh. The next step involves a nonbreath-hold CT scan. Afterward, the patient bed moves to the PET component of the scanner to complete the combined imaging process. The 1st set of data consists of nonattenuation-corrected images, which are then corrected using the CT scan images. The attenuation-corrected (AC) PET data can generate maximum intensity projection (MIP) images or be fused with CT images to provide both anatomical and functional assessments in all 3 dimensions.
The interpretation of PET/CT images primarily relies on maximum SUV (SUV max) measurements. The individual's general metabolic status influences this semiquantitative assessment. Therefore, the SUV max of the background mediastinal blood pool and liver is recorded as a reference.
No single SUV value can reliably distinguish between benign and malignant processes. As a general guideline, metabolic activity is typically categorized into 4 levels:
- Low uptake: SUV max less than 2.5
- Intermediate uptake: SUV max between 2.5 and 5
- High uptake: SUV max greater than 5
- Intense uptake: SUV max greater than 10
Complications
PET/CT is a safe procedure. The radiation dose from a routine diagnostic 18F-FDG at 350 MBq is 6 to 7 mSv. The CT component of the study has a reasonably low dose, ranging from 2 to 4 mSv if used only for attenuation correction and 3 to 10 mSv if used for both attenuation correction and localization. These values are lower than those from contrast-enhanced diagnostic CT scans, which have a dose of 15 to 25 mSv.[21]
Awareness of various false-negative and false-positive findings is crucial, as they can lead to misinterpretation. Some examples are discussed below.
False-Positive Positron Emission Tomography/Computed Tomography
Respiration or patient movement can cause a mismatch between the CT and PET data in fused images, leading to misinterpretation. This issue is especially problematic in abdominal imaging, where bowel movement can create a false impression of peritoneal disease due to misregistration of physiological bowel activity outside the bowel lumen. Nonattenuation correction (NAC) imaging can be reviewed in such situations to prevent misinterpretation.
Brown fat uptake in cold environments, particularly in the supraclavicular regions, can be misinterpreted as signals from lymph nodes. Correlating areas of uptake with CT images helps differentiate solid lesions from low Hounsfield unit fatty tissue.
Radiotracer extravasation and postvaccination status can lead to reactive lymph node uptake in the ipsilateral axilla. These events should be documented in the patient questionnaire for easy reference during reporting.
Inflammatory uptake at the site of radiotherapy, surgery, or biopsy may result in false-positive interpretations. Therefore, the timing of the scan postintervention is crucial to distinguish between malignant disease and inflammation. Chemotherapy can induce reactive marrow hyperplasia, leading to diffuse increased radiotracer uptake in the skeleton.[22]
False-Negative Positron Emission Tomography/Computed Tomography
Tumors with mucinous components may not show significant FDG uptake due to low cellularity, a lack of GLUT-1 transporters, and poor glucose metabolism.[23] Well-differentiated NETs may also show low FDG uptake. Gallium PET is preferred for assessment in such cases.
Artifacts from metallic and vascular stents, along with high activity from a distended urinary bladder, can obscure adjacent small tumors. Similarly, metformin use can lead to diffuse bowel activity, complicating the evaluation of colon pathologies (see Image. Esophagitis on Advanced Nuclear Imaging).[24]
Clinical Significance
Esophageal Cancer
After histological confirmation of malignancy, esophageal cancer is staged using the TNM classification. Early-stage tumors may undergo surgical resection with or without neoadjuvant therapy, while chemoradiotherapy followed by surgery is the standard treatment for locally advanced esophageal cancers.[25]
Esophageal cancers typically show focal or segmental areas of increased FDG uptake, contrasting with the diffuse uptake seen in esophagitis. T staging of early tumors is more accurately performed with endoscopic ultrasound, which can differentiate between the layers of the esophageal wall and assess the immediate periesophageal lymph nodes. Contrast-enhanced CT complements this approach. For nonlocoregional and distant lymph nodes, PET/CT is the most sensitive modality and plays a central role in the primary staging of FDG-avid esophageal tumors.
The 2 main histological subtypes of esophageal cancer are squamous cell carcinoma and adenocarcinoma. Squamous cell carcinoma primarily affects the upper and middle esophagus and is strongly associated with alcohol consumption and smoking. This tumor typically shows intense FDG uptake. In contrast, adenocarcinoma predominates in the lower esophagus, is linked to reflux and smoking, and exhibits highly variable FDG uptake. The incidence of adenocarcinoma is increasing, and it has become the most common subtype of esophageal cancer.
The PET/CT report should include details such as the location, length, and SUV max of the tumor, as tumor length serves as an independent prognostic factor for overall survival.[26] The most common sites of metastasis from esophageal cancer include the lungs, liver, bones, adrenal glands, and nonregional lymph nodes. Detection of occult metastasis through PET/CT, which may not be visible on conventional imaging, can help avoid unnecessary surgeries in patients who are unsuitable for curative resection (see Image. Occult Metastasis on Advanced Nuclear Imaging).
Assessing lung metastases with PET/CT is challenging due to the small size of individual lesions, which often fall below the threshold (1 cm) for PET characterization. Additionally, the nonbreath-hold CT component of the PET scan is suboptimal for assessing lung nodules. Sensitivity for liver metastases is also limited due to hepatic background activity, and if a metastasectomy is planned, preoperative magnetic resonance imaging (MRI) is essential for a more accurate assessment.
PET/CT is an excellent modality for ruling out new metastases or recurrence at distant sites following curative treatment for esophageal cancers. The sensitivity of PET/CT for detecting disease recurrence is high, with a pooled sensitivity estimate of around 96%. However, the specificity of this modality is lower, with a pooled specificity estimate of 78%, due to the potential for false-positive results caused by postoperative inflammatory changes and radiation pneumonitis.[27][27] PET/CT scans have a role in response assessment and radiotherapy planning, but concrete evidence for their routine use is lacking.[28]
Colonic Cancer
Colonic cancers can present with focal, intermediate- to high-intensity, eccentric, or short-segment radiotracer uptake. However, assessment is challenging due to several factors, including background physiological colonic uptake, bowel movement causing misregistration, and FDG uptake in fecal matter. Additionally, diffuse high colonic uptake, often seen with metformin intake, can obscure underlying pathology.[29]
Adenocarcinomas typically show variable uptake but generally exhibit high SUV max, except for mucinous adenocarcinomas, which demonstrate poor FDG avidity due to low cellularity and limited expression of GLUT-1 transporters. PET/CT is not routinely used to assess primary colorectal cancers or local lymph nodes due to the poor spatial resolution of PET photon detectors and the limited inherent contrast in the bowel.[30] The pericolonic nodes may also be obscured by the metabolic bloom of the primary tumor.[31]
MRI is typically used for the local staging of rectal cancer, while contrast-enhanced CT (CECT) is employed for staging other colonic cancers. Metastatic disease is present in about 20% of colonic cancers at diagnosis.[32] PET/CT is indicated for assessing metastatic disease in advanced tumors, particularly when resection is being contemplated. The most common sites of metastases include the liver, lungs, ovaries, and peritoneum.
Metastatic disease has been traditionally treated with systemic chemotherapy. However, recognition of metastasectomy with curative intent is increasing, supported by evidence showing improved survival rates (37%–58%) in patients who undergo surgical resection, even in the presence of multiple metastases.[33][34][35]
If surgery is considered for liver metastases, a liver MRI is essential for preoperative planning. Lung metastases require assessment with a breath-hold CT scan for precise quantification and characterization. PET/CT can detect peritoneal metastases, which may present as focal uptake or diffuse perihepatic curvilinear uptake, often corresponding with CT findings of peritoneal nodules or fat stranding. Ascites is frequently associated with peritoneal disease, although it may or may not exhibit high FDG uptake.
PET/CT plays an established role in detecting disease recurrence in patients with new clinical symptoms, abnormal examination findings, rising tumor markers, or indeterminate presacral lesions on conventional imaging. Distinguishing between recurrent disease and posttherapy changes can be challenging on PET/CT. Typically, high-intensity focal uptake persisting several months after surgery raises concern for residual or recurrent disease, while low-intensity, ill-defined uptake is expected and related to inflammatory changes.[36] PET/CT is also effective in detecting recurrence in lymph nodes and distant organs.
However, data is insufficient to definitively establish the role of PET/CT in assessing complete responders. For liver-directed therapies such as selective internal radiation therapy (SIRT), FDG-PET can be helpful, as metabolic response may be greater than morphological response.[37]
Anal Cancer
Anal cancers are rare, accounting for less than 2% of large bowel malignancies. However, their incidence has been rising in recent years, particularly in developed countries.[38] The most common cell type is squamous cell carcinoma (>70%), which is strongly associated with high-risk human papillomavirus (HPV-16) infection.[39] Combined chemoradiotherapy is curative for most patients, but those with recurrence or local failure after chemoradiotherapy may require abdominoperineal resection.
Primary anal squamous cell carcinoma exhibits very high FDG affinity, making PET/CT routinely indicated in the primary staging of anal cancer to rule out lymph nodal and hematogenous metastases (see Image. Anal Squamous Cell Carcinoma on Advanced Nuclear Imaging). Local staging is typically performed with an MRI. Additional roles of PET/CT in anal cancer management include radiotherapy planning, especially with the growing use of intensity-modulated radiation therapy (IMRT), alongside response assessment and recurrence detection.[40]
Gastrointestinal Neuroendocrine Tumors
In metastatic NETs, mixed proliferation profiles may occur at different disease sites, often becoming evident after receptor-targeted therapy. The gallium scan may show stable or regressive disease, while the CT component reveals enlargement in certain non-gallium-avid areas.[41] In such cases, FDG-PET/CT can identify increased uptake in these areas, reflecting an aggressive metabolic profile distinct from gallium-avid disease sites. This information can guide the selection of sites for tissue sampling to establish a histological correlation.
Understanding the biodistribution of 68Ga-DOTA peptides is critical for accurately interpreting pathological uptake. Normal radiotracer uptake is observed in the spleen, adrenal glands, pancreas, pituitary gland, liver, kidneys, thyroid, and salivary glands. Unlike FDG-PET/CT, 68Ga-DOTA peptide imaging shows minimal uptake in background soft tissue and muscles, enhancing the contrast of pathological findings against the background.
Enhancing Healthcare Team Outcomes
The diagnosis and management of gastrointestinal malignancies require an interprofessional approach. Upper and lower gastrointestinal endoscopies are typically the first investigations for suspected gastrointestinal malignancies. Advancements in surgery, chemotherapy, and radiotherapy have contributed to improving the overall survival rates in patients with these cancers. The judicious use of FDG-PET/CT in FDG-avid gastrointestinal malignancies plays a crucial role in reducing disease recurrence by precisely mapping disease distribution, thereby influencing treatment strategies.
Accurate stratification of patients who may benefit from curative resection has become increasingly important, as long-term survival often fails due to undetected distant metastases at the time of surgery. PET/CT's ability to detect distant disease justifies its role in the primary staging of FDG-avid gastrointestinal cancers. However, in cases where the primary tumor lacks FDG avidity, PET/CT is less reliable for assessing distant disease, as the metabolic profile of the metastases is expected to match that of the primary tumor.
PET/CT can help identify poor responders among patients undergoing neoadjuvant therapy, potentially altering management strategies for this group. However, the optimal timing for imaging and standardized response criteria for PET/CT require validation through robust evidence from randomized controlled trials.
The mechanism by which 68Ga-DOTANOC PET/CT targets SSTRs expressed by well-differentiated NETs is similarly utilized in delivering personalized molecular radiotherapy (MRT) to these tumors, a procedure known as peptide receptor radionuclide therapy (PRRT). In PRRT, gallium-68 is replaced with a therapeutic radiotracer, most commonly lutetium-177. Prior to initiating therapy, disease mapping through gallium PET or octreotide imaging determines patient suitability. Effective therapy requires that most tumor sites demonstrate radiotracer uptake above that of the liver. Following therapy, planar imaging confirms adequate radiotherapy distribution within the tumor sites.[42] This palliative therapy is being extensively studied to evaluate survival benefits in selected patient populations, offering a promising option for managing advanced neuroendocrine tumors.
Media
(Click Image to Enlarge)
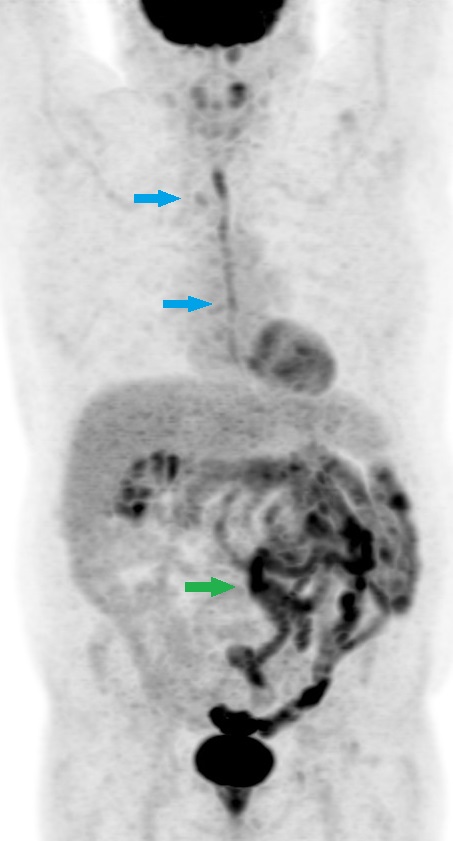
Esophagitis on Advanced Nuclear Imaging. Fluorodeoxyglucose positron emission tomography/computed tomography in a patient with dysphagia shows diffuse increased radiotracer uptake in the esophagus consistent with esophagitis (blue arrows). Note that heterogeneous uptake in the colon (green arrow) is due to metformin intake.
Contributed by Dr Amaila Ramzan, FRCR
(Click Image to Enlarge)
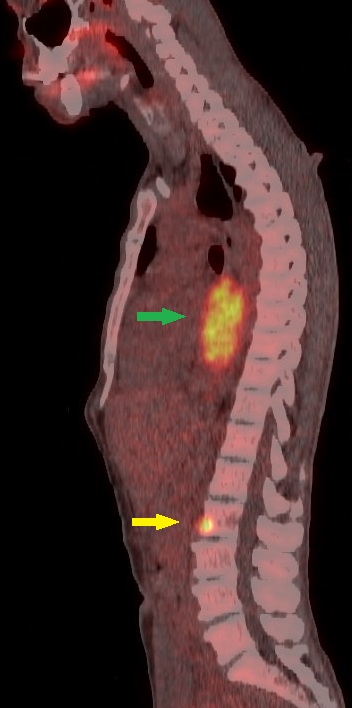
Occult Metastasis on Advanced Nuclear Imaging. This sagittal fused fluorodeoxyglucose positron emission tomography/computed tomography (FDG PET/CT) image of a 56-year-old patient shows a highly FDG-avid (SUV max 18) mass of the midthoracic esophagus (green arrow) with solitary metastasis in the L2 vertebral body (yellow arrow). FDG PET/CT identified a metastatic lesion that was not detected on the staging CT scan, resulting in an upstaging of the disease.
Contributed by Dr Amaila Ramzan, FRCR
(Click Image to Enlarge)
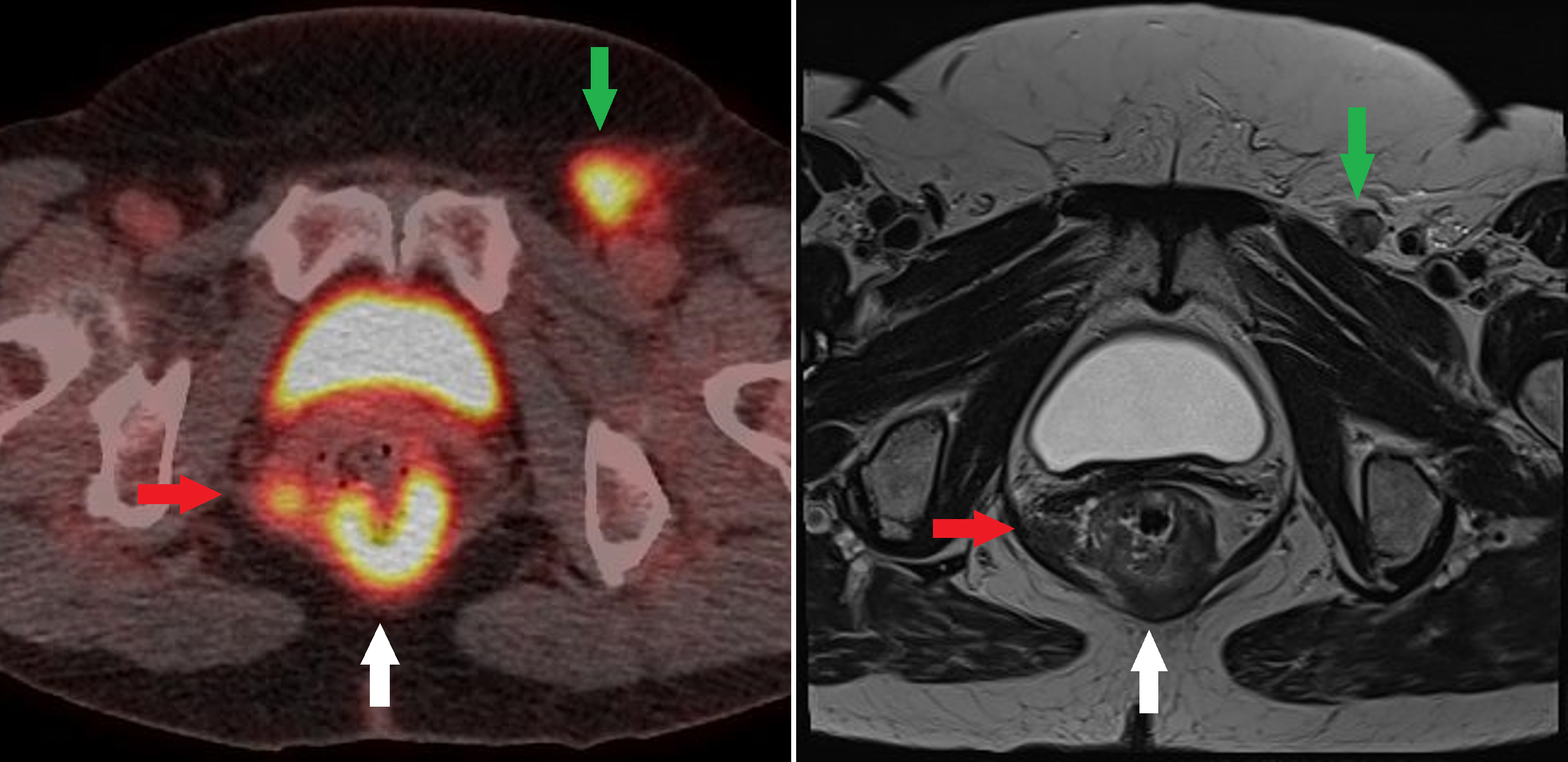
Anal Squamous Cell Carcinoma. Fused fluorodeoxyglucose positron emission tomography/computed tomography and T2 axial magnetic resonance images at the same level of an anorectal junction tumor (white arrow) with a fluorodeoxyglucose-avid nodal deposit in the right mesorectum (red arrow) and the left groin (green arrow).
Contributed by A Ramzan, FRCR
(Click Image to Enlarge)
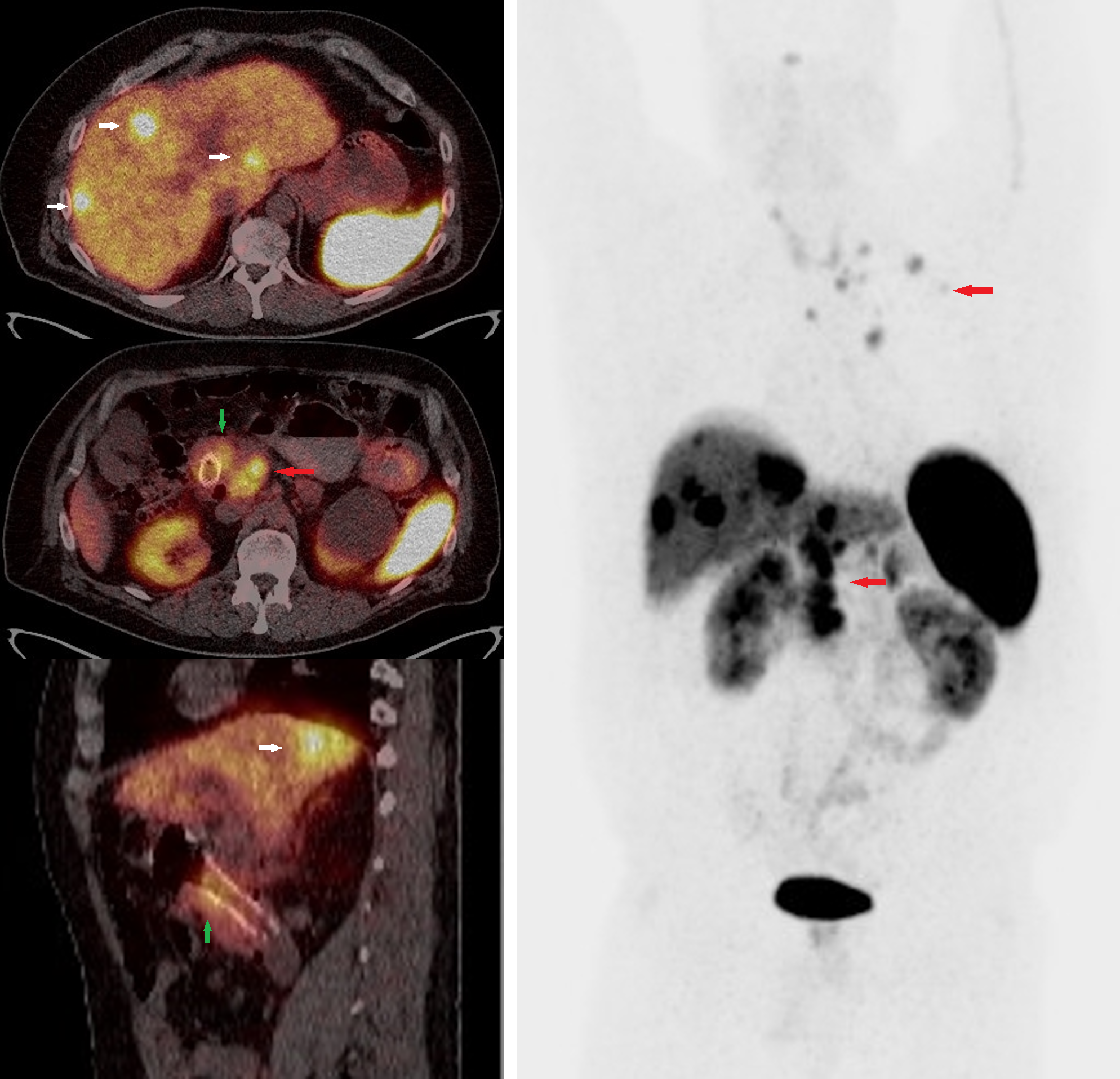
Gastrointestinal Neuroendocrine Tumor on Advanced Nuclear Imaging. Gallum-68 DOTATOC positron emission tomography/computed tomography whole-body scan fused and maximum intensity projection images of a patient with well-differentiated grade 2 neuroendocrine tumor of the duodenum with metastatic disease to the liver and lymph nodes. The green arrow shows inflammatory uptake around the stent in the duodenal tumor. The red arrows show radiotracer uptake in conglomerate retroperitoneal and thoracic lymph nodes. The white arrow shows radiotracer avid hepatic metastases.
Contributed by Dr Amaila Ramzan, FRCR
References
Bentley-Hibbert S, Schwartz L. Use of Imaging for GI Cancers. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2015 Jun 1:33(16):1729-36. doi: 10.1200/JCO.2014.60.2847. Epub 2015 Apr 27 [PubMed PMID: 25918300]
Vaupel P, Schmidberger H, Mayer A. The Warburg effect: essential part of metabolic reprogramming and central contributor to cancer progression. International journal of radiation biology. 2019 Jul:95(7):912-919. doi: 10.1080/09553002.2019.1589653. Epub 2019 Mar 22 [PubMed PMID: 30822194]
Özgüven S, Filizoğlu N, Kesim S, Öksüzoğlu K, Şen F, Öneş T, İnanır S, Turoğlu HT, Erdil TY. Physiological Biodistribution of (68)Ga-DOTA-TATE in Normal Subjects. Molecular imaging and radionuclide therapy. 2021 Feb 9:30(1):39-46. doi: 10.4274/mirt.galenos.2021.37268. Epub [PubMed PMID: 33586406]
Münch S, Marr L, Feuerecker B, Dapper H, Braren R, Combs SE, Duma MN. Impact of (18)F-FDG-PET/CT on the identification of regional lymph node metastases and delineation of the primary tumor in esophageal squamous cell carcinoma patients. Strahlentherapie und Onkologie : Organ der Deutschen Rontgengesellschaft ... [et al]. 2020 Sep:196(9):787-794. doi: 10.1007/s00066-020-01630-y. Epub 2020 May 19 [PubMed PMID: 32430661]
Shepherd NA, Baxter KJ, Love SB. The prognostic importance of peritoneal involvement in colonic cancer: a prospective evaluation. Gastroenterology. 1997 Apr:112(4):1096-102 [PubMed PMID: 9097991]
Salati SA, Al Kadi A. Anal cancer - a review. International journal of health sciences. 2012 Jun:6(2):206-30 [PubMed PMID: 23580899]
Klimstra DS, Modlin IR, Coppola D, Lloyd RV, Suster S. The pathologic classification of neuroendocrine tumors: a review of nomenclature, grading, and staging systems. Pancreas. 2010 Aug:39(6):707-12. doi: 10.1097/MPA.0b013e3181ec124e. Epub [PubMed PMID: 20664470]
Cives M, Strosberg JR. Gastroenteropancreatic Neuroendocrine Tumors. CA: a cancer journal for clinicians. 2018 Nov:68(6):471-487. doi: 10.3322/caac.21493. Epub 2018 Oct 8 [PubMed PMID: 30295930]
Uenomachi M, Takahashi M, Shimazoe K, Takahashi H, Kamada K, Orita T, Ogane K, Tsuji AB. Simultaneous in vivo imaging with PET and SPECT tracers using a Compton-PET hybrid camera. Scientific reports. 2021 Sep 9:11(1):17933. doi: 10.1038/s41598-021-97302-7. Epub 2021 Sep 9 [PubMed PMID: 34504184]
Karfis I, Marin G, Levillain H, Drisis S, Muteganya R, Critchi G, Taraji-Schiltz L, Guix CA, Shaza L, Elbachiri M, Mans L, Machiels G, Hendlisz A, Flamen P. Prognostic value of a three-scale grading system based on combining molecular imaging with (68)Ga-DOTATATE and (18)F-FDG PET/CT in patients with metastatic gastroenteropancreatic neuroendocrine neoplasias. Oncotarget. 2020 Feb 11:11(6):589-599. doi: 10.18632/oncotarget.27460. Epub 2020 Feb 11 [PubMed PMID: 32110279]
Long NM, Smith CS. Causes and imaging features of false positives and false negatives on F-PET/CT in oncologic imaging. Insights into imaging. 2011 Dec:2(6):679-698 [PubMed PMID: 22347986]
Hicks RJ, Binns D, Stabin MG. Pattern of uptake and excretion of (18)F-FDG in the lactating breast. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2001 Aug:42(8):1238-42 [PubMed PMID: 11483686]
Boellaard R, Delgado-Bolton R, Oyen WJ, Giammarile F, Tatsch K, Eschner W, Verzijlbergen FJ, Barrington SF, Pike LC, Weber WA, Stroobants S, Delbeke D, Donohoe KJ, Holbrook S, Graham MM, Testanera G, Hoekstra OS, Zijlstra J, Visser E, Hoekstra CJ, Pruim J, Willemsen A, Arends B, Kotzerke J, Bockisch A, Beyer T, Chiti A, Krause BJ, European Association of Nuclear Medicine (EANM). FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. European journal of nuclear medicine and molecular imaging. 2015 Feb:42(2):328-54. doi: 10.1007/s00259-014-2961-x. Epub 2014 Dec 2 [PubMed PMID: 25452219]
Level 1 (high-level) evidenceBenamor M, Ollivier L, Brisse H, Moulin-Romsee G, Servois V, Neuenschwander S. PET/CT imaging: what radiologists need to know. Cancer imaging : the official publication of the International Cancer Imaging Society. 2007 Oct 1:7 Spec No A(Special issue A):S95-9 [PubMed PMID: 17921089]
Nadig V, Herrmann K, Mottaghy FM, Schulz V. Hybrid total-body pet scanners-current status and future perspectives. European journal of nuclear medicine and molecular imaging. 2022 Jan:49(2):445-459. doi: 10.1007/s00259-021-05536-4. Epub 2021 Oct 14 [PubMed PMID: 34647154]
Level 3 (low-level) evidenceVaquero JJ, Kinahan P. Positron Emission Tomography: Current Challenges and Opportunities for Technological Advances in Clinical and Preclinical Imaging Systems. Annual review of biomedical engineering. 2015:17():385-414. doi: 10.1146/annurev-bioeng-071114-040723. Epub [PubMed PMID: 26643024]
Level 3 (low-level) evidenceBoellaard R, O'Doherty MJ, Weber WA, Mottaghy FM, Lonsdale MN, Stroobants SG, Oyen WJ, Kotzerke J, Hoekstra OS, Pruim J, Marsden PK, Tatsch K, Hoekstra CJ, Visser EP, Arends B, Verzijlbergen FJ, Zijlstra JM, Comans EF, Lammertsma AA, Paans AM, Willemsen AT, Beyer T, Bockisch A, Schaefer-Prokop C, Delbeke D, Baum RP, Chiti A, Krause BJ. FDG PET and PET/CT: EANM procedure guidelines for tumour PET imaging: version 1.0. European journal of nuclear medicine and molecular imaging. 2010 Jan:37(1):181-200. doi: 10.1007/s00259-009-1297-4. Epub [PubMed PMID: 19915839]
Ozaslan E, Kiziltepe M, Addulrezzak U, Kula M, Bozkurt O, Kut E, Duran AO, Ucar M, Sakalar T, Dogan E, Topaloglu US, Inanc M, Ozkan M. Is SUVmax of (18)F-FDG PET/CT Predictive Factor for Malignancy in Gastrointestinal Tract? Nigerian journal of clinical practice. 2021 Aug:24(8):1217-1224. doi: 10.4103/njcp.njcp_637_18. Epub [PubMed PMID: 34397034]
Shirone N, Shinkai T, Yamane T, Uto F, Yoshimura H, Tamai H, Imai T, Inoue M, Kitano S, Kichikawa K, Hasegawa M. Axillary lymph node accumulation on FDG-PET/CT after influenza vaccination. Annals of nuclear medicine. 2012 Apr:26(3):248-52. doi: 10.1007/s12149-011-0568-x. Epub 2012 Jan 21 [PubMed PMID: 22271546]
Level 2 (mid-level) evidenceDelbeke D, Coleman RE, Guiberteau MJ, Brown ML, Royal HD, Siegel BA, Townsend DW, Berland LL, Parker JA, Hubner K, Stabin MG, Zubal G, Kachelriess M, Cronin V, Holbrook S. Procedure guideline for tumor imaging with 18F-FDG PET/CT 1.0. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2006 May:47(5):885-95 [PubMed PMID: 16644760]
Level 1 (high-level) evidenceLeide-Svegborn S. Radiation exposure of patients and personnel from a PET/CT procedure with 18F-FDG. Radiation protection dosimetry. 2010 Apr-May:139(1-3):208-13. doi: 10.1093/rpd/ncq026. Epub 2010 Feb 18 [PubMed PMID: 20167792]
Griffeth LK. Use of PET/CT scanning in cancer patients: technical and practical considerations. Proceedings (Baylor University. Medical Center). 2005 Oct:18(4):321-30 [PubMed PMID: 16252023]
Berger KL, Nicholson SA, Dehdashti F, Siegel BA. FDG PET evaluation of mucinous neoplasms: correlation of FDG uptake with histopathologic features. AJR. American journal of roentgenology. 2000 Apr:174(4):1005-8 [PubMed PMID: 10749239]
Level 2 (mid-level) evidenceSteenkamp DW, McDonnell ME, Meibom S. Metformin may be associated with false-negative cancer detection in the gastrointestinal tract on PET/CT. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. 2014 Oct:20(10):1079-83. doi: 10.4158/EP14127.RA. Epub [PubMed PMID: 25100379]
Lordick F, Mariette C, Haustermans K, Obermannová R, Arnold D, ESMO Guidelines Committee. Oesophageal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of oncology : official journal of the European Society for Medical Oncology. 2016 Sep:27(suppl 5):v50-v57 [PubMed PMID: 27664261]
Level 1 (high-level) evidenceHollis AC, Quinn LM, Hodson J, Evans E, Plowright J, Begum R, Mitchell H, Hallissey MT, Whiting JL, Griffiths EA. Prognostic significance of tumor length in patients receiving esophagectomy for esophageal cancer. Journal of surgical oncology. 2017 Dec:116(8):1114-1122. doi: 10.1002/jso.24789. Epub 2017 Aug 2 [PubMed PMID: 28767142]
Goense L, van Rossum PS, Reitsma JB, Lam MG, Meijer GJ, van Vulpen M, Ruurda JP, van Hillegersberg R. Diagnostic Performance of ¹⁸F-FDG PET and PET/CT for the Detection of Recurrent Esophageal Cancer After Treatment with Curative Intent: A Systematic Review and Meta-Analysis. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2015 Jul:56(7):995-1002. doi: 10.2967/jnumed.115.155580. Epub 2015 May 7 [PubMed PMID: 25952733]
Level 1 (high-level) evidenceKitajima K, Nakajo M, Kaida H, Minamimoto R, Hirata K, Tsurusaki M, Doi H, Ueno Y, Sofue K, Tamaki Y, Yamakado K. Present and future roles of FDG-PET/CT imaging in the management of gastrointestinal cancer: an update. Nagoya journal of medical science. 2017 Nov:79(4):527-543. doi: 10.18999/nagjms.79.4.527. Epub [PubMed PMID: 29238109]
Jayaprakasam VS, Paroder V, Schöder H. Variants and Pitfalls in PET/CT Imaging of Gastrointestinal Cancers. Seminars in nuclear medicine. 2021 Sep:51(5):485-501. doi: 10.1053/j.semnuclmed.2021.04.001. Epub 2021 May 6 [PubMed PMID: 33965198]
Moses WW. Fundamental Limits of Spatial Resolution in PET. Nuclear instruments & methods in physics research. Section A, Accelerators, spectrometers, detectors and associated equipment. 2011 Aug 21:648 Supplement 1():S236-S240 [PubMed PMID: 21804677]
Dahmarde H, Parooie F, Salarzaei M. Is (18)F-FDG PET/CT an Accurate Way to Detect Lymph Node Metastasis in Colorectal Cancer: A Systematic Review and Meta-Analysis. Contrast media & molecular imaging. 2020:2020():5439378. doi: 10.1155/2020/5439378. Epub 2020 Jul 18 [PubMed PMID: 32733174]
Level 1 (high-level) evidenceRiihimäki M, Hemminki A, Sundquist J, Hemminki K. Patterns of metastasis in colon and rectal cancer. Scientific reports. 2016 Jul 15:6():29765. doi: 10.1038/srep29765. Epub 2016 Jul 15 [PubMed PMID: 27416752]
Choti MA, Sitzmann JV, Tiburi MF, Sumetchotimetha W, Rangsin R, Schulick RD, Lillemoe KD, Yeo CJ, Cameron JL. Trends in long-term survival following liver resection for hepatic colorectal metastases. Annals of surgery. 2002 Jun:235(6):759-66 [PubMed PMID: 12035031]
Level 2 (mid-level) evidenceFernandez FG, Drebin JA, Linehan DC, Dehdashti F, Siegel BA, Strasberg SM. Five-year survival after resection of hepatic metastases from colorectal cancer in patients screened by positron emission tomography with F-18 fluorodeoxyglucose (FDG-PET). Annals of surgery. 2004 Sep:240(3):438-47; discussion 447-50 [PubMed PMID: 15319715]
Saito Y, Omiya H, Kohno K, Kobayashi T, Itoi K, Teramachi M, Sasaki M, Suzuki H, Takao H, Nakade M. Pulmonary metastasectomy for 165 patients with colorectal carcinoma: A prognostic assessment. The Journal of thoracic and cardiovascular surgery. 2002 Nov:124(5):1007-13 [PubMed PMID: 12407386]
Even-Sapir E, Parag Y, Lerman H, Gutman M, Levine C, Rabau M, Figer A, Metser U. Detection of recurrence in patients with rectal cancer: PET/CT after abdominoperineal or anterior resection. Radiology. 2004 Sep:232(3):815-22 [PubMed PMID: 15273334]
Level 2 (mid-level) evidenceSzyszko T, Al-Nahhas A, Canelo R, Habib N, Jiao L, Wasan H, Pagou M, Tait P. Assessment of response to treatment of unresectable liver tumours with 90Y microspheres: value of FDG PET versus computed tomography. Nuclear medicine communications. 2007 Jan:28(1):15-20 [PubMed PMID: 17159544]
Grulich AE, Poynten IM, Machalek DA, Jin F, Templeton DJ, Hillman RJ. The epidemiology of anal cancer. Sexual health. 2012 Dec:9(6):504-8. doi: 10.1071/SH12070. Epub [PubMed PMID: 22958581]
Shiels MS, Kreimer AR, Coghill AE, Darragh TM, Devesa SS. Anal Cancer Incidence in the United States, 1977-2011: Distinct Patterns by Histology and Behavior. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2015 Oct:24(10):1548-56. doi: 10.1158/1055-9965.EPI-15-0044. Epub 2015 Jul 29 [PubMed PMID: 26224796]
Di Carlo C, di Benedetto M, Vicenzi L, Costantini S, Cucciarelli F, Fenu F, Arena E, Mariucci C, Montisci M, Panni V, Patani F, Valenti M, Palucci A, Burroni L, Mantello G. FDG-PET/CT in the Radiotherapy Treatment Planning of Locally Advanced Anal Cancer: A Monoinstitutional Experience. Frontiers in oncology. 2021:11():655322. doi: 10.3389/fonc.2021.655322. Epub 2021 Jul 1 [PubMed PMID: 34277406]
Tang LH, Untch BR, Reidy DL, O'Reilly E, Dhall D, Jih L, Basturk O, Allen PJ, Klimstra DS. Well-Differentiated Neuroendocrine Tumors with a Morphologically Apparent High-Grade Component: A Pathway Distinct from Poorly Differentiated Neuroendocrine Carcinomas. Clinical cancer research : an official journal of the American Association for Cancer Research. 2016 Feb 15:22(4):1011-7. doi: 10.1158/1078-0432.CCR-15-0548. Epub 2015 Oct 19 [PubMed PMID: 26482044]
Pobłocki J, Jasińska A, Syrenicz A, Andrysiak-Mamos E, Szczuko M. The Neuroendocrine Neoplasms of the Digestive Tract: Diagnosis, Treatment and Nutrition. Nutrients. 2020 May 15:12(5):. doi: 10.3390/nu12051437. Epub 2020 May 15 [PubMed PMID: 32429294]