Summary / Explanation
Introduction
What was once considered a painful procedure has become routine with the advent of local anesthesia. Local anesthetic drugs work by reversibly blocking sodium channels, preventing the influx of sodium into the cell. This inhibition of depolarization and the transmission of action potentials enables the performance of dental procedures, such as drilling into the dentin, pain-free and with minimal discomfort.[1][2]
Pharmacology
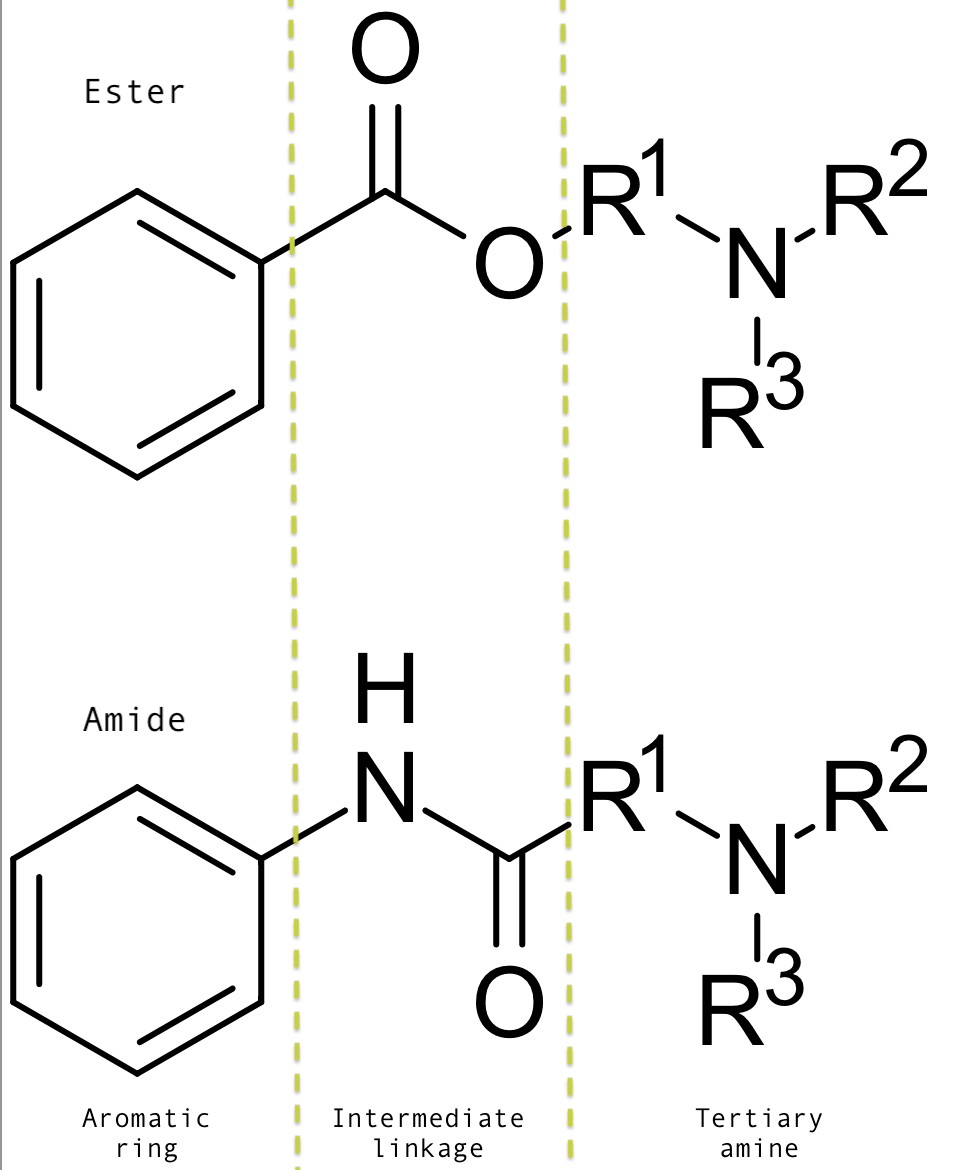
Two main classes of local anesthetic drugs are used in dentistry. They are classified based on their chemical structure into amides and esters. Both share the same mechanism of action but differ in their duration of action and how they are metabolized. Ester-type agents have a shorter duration of action of around 15 minutes and are metabolized via plasma cholinesterase in the bloodstream. Although articaine belongs to the amide-type local anesthetics, it is also metabolized via plasma esterases due to having an additional ester linkage.[1] Amide local anesthetics have a considerably longer duration of action and are metabolized in the liver via microsomal enzymes.[1][3]
The structure of local anesthetics is divided into three main components: an aromatic ring, an intermediate ester or amide, and a terminal amine (see Image. Structure of ester and amide local anesthesia agents). The aromatic ring is lipophilic, promoting the diffusion of the anesthetic across the neural membrane: the more lipophilic, the greater the potency. The terminal amine is arranged in either a lipid-soluble tertiary manner or a water-soluble quaternary manner, enabling active and inactive states of the local anesthetic.
Most local anesthetics commonly used in dental procedures belong to the amide group, such as lidocaine, articaine, mepivacaine, and bupivacaine. Procaine, benzocaine, and cocaine belong to the ester group.[2] Benzocaine is mainly used as a topical anesthesia agent prior to injection.
Cocaine was the first local anesthetic to be discovered. Cocaine had good efficacy, but it came out of use due to undesirable adverse effects, such as cardiac and CNS stimulation, euphoria, and addictive potential. Furthermore, cocaine had a higher incidence of CNS and cardiovascular toxicity, making it inappropriate for medical use. Procaine came into use in 1905, gaining popularity in the United States; however, it was found to have a high potential for allergic reactions.[2]
Local Anesthetic Agents
Lidocaine
Lidocaine is the most widely used local anesthetic today. Lidocaine has the longest track record for safety and efficacy in routine dental procedures and is regarded as the gold standard against which all new local anesthetic drugs are evaluated. Belonging to the amide class, it contains a benzene aromatic ring and is associated with a rare incidence of life-threatening hypersensitivity reactions. However, neurotoxicity may occur if used at high concentrations.[3]
Lidocaine is typically combined with the vasoconstrictor adrenaline, which stimulates α1-adrenergic receptors in arteriole walls. Adrenaline prolongs the anesthesia and enables a lower concentration of local anesthetic to be delivered. Lidocaine induces vasoconstriction in the area of infiltration, reducing the rate of plasma uptake of the local anesthetic agent and the potential for toxicity. Additionally, vasoconstriction aids hemostasis to provide a cleaner procedural field.[4] Even at the low systemic concentrations used in dentistry, adrenaline may increase heart rate, cardiac output, and peripheral vasodilation. These effects are amplified in medically compromised patients, with the potential to trigger a hypertensive crisis, myocardial infarction, or arrhythmia.[5]
Metabolism of lidocaine principally occurs in the liver, with around 10% being excreted unaltered via urine. The elimination half-life is estimated to be around 90 minutes. This half-life may be extended in patients with advanced liver disease. Lower doses of anesthetic drugs should be considered for patients with liver or kidney disease, as deteriorated function impacts drug metabolism and clearance.[6]
Prilocaine
Prilocaine is associated with lower toxicity and vasodilation than lidocaine. It is often used for patients where a vasoconstrictor is contraindicated, such as those with unstable angina and uncontrolled severe hypertension. Prilocaine is typically manufactured in combination with felypressin, a catecholamine vasoconstrictor chemically similar to vasopressin. Felypressin reduces the rate of venous outflow and is less vasoconstrictive than adrenaline.[7] Prilocaine's reduced vasodilation overcomes the less vasoconstrictive effect of felypressin. Thus, the duration of action of prilocaine is prolonged, and adequate anesthesia can be achieved. Felypressin is best avoided during pregnancy as it mimics the effect of oxytocin and may potentially induce uterine contractions.[3] Prilocaine is considered less toxic than lidocaine as it is partly metabolized in the lungs and kidneys, placing less burden on the liver. In addition, it clears from the kidneys more rapidly than other amides.[3]
Articaine
First synthesized in 1969, articaine has gained rapid popularity globally. Articaine is a unique amide containing thiophene instead of a benzene ring. The thiophene ring improves lipid solubility and diffusion through the lipid membrane of neurons.[8] This enhanced solubility, combined with the higher concentration of 4% in which it is manufactured, improves the diffusion of anesthesia in the mandible.
Articaine has an efficient pattern of clearance. Once absorbed into the bloodstream from the injection site, articaine is distributed throughout body tissues. The thiophene ring contains an ester side chain inactivated by plasma esterases in the blood.[9] Articaine has a half-life of around 20 minutes, which is much shorter than lidocaine and other amides hepatically metabolized.
Articaine has low toxicity, which makes it safe for lengthy procedures when prolonged anesthesia is required.[10] Articaine's ester group allows approximately 90% of articaine to be metabolized into articainic acid in the plasma, subsequently excreted by the kidneys. The remaining 10% undergoes metabolism in the liver.
Gulenko et al. found articaine to be an effective and safe alternative to lidocaine for children under 4 years of age, supporting its use in pediatric patients of all ages. The recommended dose for children is 5 mg/kg, which should be carefully calculated at each appointment. While studies support the safety of articaine in pediatric patients, caution is advised when administering it to children aged 4 and below.[11]
The use of articaine in dentistry has been extensively studied. While concerns have been raised about its potential neurotoxicity—particularly reports of paraesthesia following inferior alveolar nerve blocks and prolonged numbness after dental procedures—numerous studies have concluded that articaine is a safe and effective local anesthetic for patients of all ages in dental practice.[12]
Mepivacaine
Mepivacaine is an essential amide local anesthetic in dentistry, known for its rapid onset and moderate duration of action. Similar to prilocaine, mepivacaine has less vasodilating activity than lidocaine. This property allows it to be used without a vasoconstrictor, allowing the administration of higher concentrations of plain 3% mepivacaine directly at the site.[13] As a result, it serves as a practical alternative to lidocaine or articaine combined with adrenaline.
Mepivacaine is often preferred for medically compromised patients because it does not require a vasoconstrictor, minimizing the risk of increased heart rate and blood pressure. This makes it an ideal choice for older patients with cardiovascular disease and individuals with hyperthyroidism.[14] However, the anesthesia effect of mepivacaine is less potent than lidocaine.
The onset of local anesthesia is influenced by its ionization constant (pKa) and lipid solubility. Local anesthetics are formulated as hydrochloride salts to ensure their stability in the solution. In the water-soluble state, they cannot cross nerve membranes. Adequate anesthesia occurs when anesthetic molecules convert to a lipid-soluble state, a process dependent on physiologic pH. Local anesthetics with a higher pKa have fewer molecules in the lipid-soluble state, leading to slower onset times. This conversion is further limited in infected tissues, where the environment is more acidic.[1]
Mepivacaine has an advantage in cases of inflammation or infection due to its higher pH and lower pKa, enabling it to cross nerve membranes more effectively in acidic environments. Consequently, it may provide superior anesthesia than lidocaine in such cases. Older patients frequently suffer from conditions affecting reserve capacity and renal and hepatic function. Therefore, limiting the overall dose of local anesthetic is generally recommended to reduce the risk of toxicity. Additionally, cardiac conditions may impair the clearance of local anesthetics, further supporting the need for dose adjustments.
Bupivacaine
Bupivacaine is a water-soluble, long-acting amide anesthetic. Bupivacaine is particularly beneficial for postoperative pain management due to its prolonged duration of anesthesia, which is attributed to its high lipid solubility and strong protein-binding potential. Bupivacaine provides adequate anesthesia for pulp, bone, and soft tissues, making it suitable for many dental procedures, particularly lengthy treatments such as root canal treatment.[15]
The onset of anesthesia with bupivacaine is slower than that of other local anesthetics, typically taking 5 to 8 minutes in both the maxilla and mandible. In contrast, lidocaine provides a faster onset, usually within 2 to 4 minutes. To address this delay, a common approach is to administer lidocaine initially for rapid onset, followed by a bupivacaine top-up to ensure prolonged anesthesia for both operative and postoperative pain management.[16]
While bupivacaine's long-lasting effects are advantageous for patient comfort during and after procedures, prolonged soft tissue anesthesia is undesirable in some instances. Bupivacaine is, therefore, reserved for lengthy procedures likely to result in significant postoperative pain.[11] Research by Malamed indicates that the duration of soft tissue anesthesia with 0.5% bupivacaine combined with 1:200,000 adrenaline ranges from 4 to 9 hours, compared to 3 to 5 hours for 2% lidocaine with 1:100,000 adrenaline.[15] However, this prolonged tissue anesthesia may not be ideal for specific patient groups, such as children or individuals with special needs, where the risk of accidental soft tissue biting is higher.
Although the incidence of toxicity from local anesthetics is rare, bupivacaine has a higher cardiotoxicity compared to other agents. Toxic levels of bupivacaine can affect cardiac tissues, impairing conduction, cardiac contractility, and vascular tone due to significant vasodilation.[15]
Topical Anesthetics
Topical anesthetics act on peripheral nerves to minimize the discomfort of needle insertion or eliminate its need in certain cases. They may also be applied to the soft palate to reduce gag reflexes associated with impressions or intraoral radiographs. Topical agents come in the form of gels, sprays, and ointments. Their concentration is higher than local anesthetics, which helps promote diffusion across superficial mucosal layers. The area is dried, and the agent is applied using a spay or cotton swab.
Benzocaine formulations present as an ointment, spray, or gel with concentrations ranging from 6% to 20%. Ointment preparations can be beneficial in providing relief for patients with aphthous ulcers. At a concentration of 20%, benzocaine works within 30 seconds with adequate depth of topical anesthesia achieved at 2 to 3 minutes. Lidocaine is also available as a 2% or 5% gel or a 10% spray. The onset of action is around 1 to 2 minutes, with adequate anesthesia effect after 3 minutes.[17]
Maximum Dose
Local anesthetics have the potential to cause toxicity if the maximum dose is exceeded, leading to adverse neurological and cardiac effects. Toxicity results from neural ischemia and inflammation in response to the anesthetic drug or vasoconstrictor. Early signs include excitatory neurological symptoms such as visual and sensory disturbances, as well as seizures.[18] These may progress to depressive symptoms, including reduced consciousness, coma, or respiratory failure.[18] Cardiac effects may range from tachyarrhythmia or bradyarrhythmia to cardiac arrest.
Preventing toxicity involves adhering to the safe dosage limits based on the patient's weight. The concentration of the anesthetic solution is expressed as a percentage, representing grams of anesthetic per 100 ml. For example, for lidocaine, a 2% solution equals 20 mg/ml (2 grams of lidocaine per 100 ml).[18]
To calculate the total anesthetic dose in a cartridge, the solution's concentration in mg/ml is multiplied by the volume of the cartridge, commonly 1.8 ml or 2.2 ml (20mg/ml x 2.2 ml = 44 mg). A healthy adult weighing 70 kg can have a maximum of 490 mg of lidocaine with vasoconstrictor (7mg/kg x 70 kg = 490 mg) (See Table 1).[18] To calculate the maximum number of cartridges for this individual, the maximum dose of the drug is divided by the mg in each cartridge (490 mg/44 mg = 11.13 cartridges.[18] The same calculation can be used to calculate the maximum number of cartridges based on different weights and anesthetic agents.
Potency, dose, and time of exposure to a local anesthetic are important factors for the development of toxicity. Children or frail patients may require below-average dosages. Clinical signs of toxicity include slurred speech, diplopia, tinnitus, and muscle twitching. Acute toxicity may be caused by accidental intravascular administration. Recommendations for maximum doses of local anesthetics are in Table 1. Generally, the site of local anesthetic injection in conjunction with patient-related factors is important to evaluate when considering the dose.[18]
Table 1. Maximum Local Anesthetic Doses [1]
|
Anesthetic |
Maximum Dose |
|
4% Articaine with 1:100,000 adrenaline |
7 mg/kg (up to 500 mg) 5 mg/kg in children |
|
2% Lidocaine with 1:80,000 adrenaline or 1:100,000 adrenaline |
7 mg/kg (up to 500 mg) |
|
4% Prilocaine with 1:200,000 adrenaline |
8 mg/kg (up to 500 mg) |
|
0.5% Bupivacaine with 1:200,000 adrenaline |
2 mg/kg (up to 200 mg) |
|
3% Mepivacaine |
6.6 mg/kg (up to 400 mg) |
Computer-Controlled Local Anesthesia Delivery (CCLAD) Systems:
In recent years, significant advancements have been made to reduce the discomfort associated with administering local anesthesia, which can be particularly beneficial for children and individuals with dental fear and anxiety. Discomfort during injection can result from factors such as tissue perforation, pressure from solution dispersion, and the temperature and pH of the anesthetic solution. CCLAD systems work to overcome this issue by delivering the local anesthetic solution at a consistent slow rate and pressure. This can make the injection almost imperceptible.[19]
These systems are composed of a computer connected to a pen-like handpiece. This has a less threatening appearance than a conventional syringe and facilitates precision when inserting the needle. However, drawbacks of the system include higher cost and space requirements of the equipment and the increased time taken to administer the local anesthetic drug.[20]
Conclusion
Local anesthetics are pivotal to modern dentistry. They allow pain-free and comfortable procedures and reinforce positive relationships between dentists and patients. Advancements in local anesthetic drugs have significantly improved their safety, efficacy, and versatility. Understanding the pharmacology, duration of action, and potential toxicity is crucial for optimizing patient care.
The selection of a local anesthetic should be guided by each patient's specific needs, the procedure's type and duration, and postoperative pain management considerations. Furthermore, careful evaluation of the patient's medical history, such as cardiovascular diseases, is vital. Including or excluding a vasoconstrictor must balance its benefits, such as prolonged anesthesia and hemostasis, against potential systemic effects. Tailoring the choice of local anesthetics to each case improves overall treatment outcomes.
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Media
(Click Image to Enlarge)
References
Decloux D, Ouanounou A. Local anaesthesia in dentistry: a review. International dental journal. 2020 Sep 17:71(2):87-95. doi: 10.1111/idj.12615. Epub 2020 Sep 17 [PubMed PMID: 32944974]
Sheikh NK, Dua A. Procaine. StatPearls. 2025 Jan:(): [PubMed PMID: 31855383]
Gazal G, Omar E, Elmalky W. Rules of selection for a safe local anesthetic in dentistry. Journal of Taibah University Medical Sciences. 2023 Dec:18(6):1195-1196. doi: 10.1016/j.jtumed.2023.04.007. Epub 2023 May 3 [PubMed PMID: 37234724]
Karm MH, Park FD, Kang M, Kim HJ, Kang JW, Kim S, Kim YD, Kim CH, Seo KS, Kwon KH, Kim CH, Lee JW, Hong SW, Lim MH, Nam SK, Cho JM. Comparison of the efficacy and safety of 2% lidocaine HCl with different epinephrine concentration for local anesthesia in participants undergoing surgical extraction of impacted mandibular third molars: A multicenter, randomized, double-blind, crossover, phase IV trial. Medicine. 2017 May:96(21):e6753. doi: 10.1097/MD.0000000000006753. Epub [PubMed PMID: 28538371]
Level 1 (high-level) evidenceTorres-Lagares D, Serrera-Figallo MÁ, Machuca-Portillo G, Corcuera-Flores JR, Machuca-Portillo C, Castillo-Oyagüe R, Gutiérrez-Pérez JL. Cardiovascular effect of dental anesthesia with articaine (40 mg with epinefrine 0,5 mg % and 40 mg with epinefrine 1 mg%) versus mepivacaine (30 mg and 20 mg with epinefrine 1 mg%) in medically compromised cardiac patients: a cross-over, randomized, single blinded study. Medicina oral, patologia oral y cirugia bucal. 2012 Jul 1:17(4):e655-60 [PubMed PMID: 22322521]
Level 1 (high-level) evidenceTorp KD, Metheny E, Simon LV. Lidocaine Toxicity. StatPearls. 2025 Jan:(): [PubMed PMID: 29494086]
Gazal G. Is prilocaine safe and potent enough for use in the oral surgery of medically compromised patients. Saudi medical journal. 2019 Jan:40(1):97-100. doi: 10.15537/smj.2019.01.23475. Epub [PubMed PMID: 30617388]
Martin E, Nimmo A, Lee A, Jennings E. Articaine in dentistry: an overview of the evidence and meta-analysis of the latest randomised controlled trials on articaine safety and efficacy compared to lidocaine for routine dental treatment. BDJ open. 2021 Jul 17:7(1):27. doi: 10.1038/s41405-021-00082-5. Epub 2021 Jul 17 [PubMed PMID: 34274944]
Level 1 (high-level) evidenceDarawade DA, Kumar S, Budhiraja S, Mittal M, Mehta TN. A clinical study of efficacy of 4% articaine hydrochloride versus 2% lignocaine hydrochloride in dentistry. Journal of international oral health : JIOH. 2014 Sep:6(5):81-3 [PubMed PMID: 25395799]
Becker DE, Reed KL. Local anesthetics: review of pharmacological considerations. Anesthesia progress. 2012 Summer:59(2):90-101; quiz 102-3. doi: 10.2344/0003-3006-59.2.90. Epub [PubMed PMID: 22822998]
Luo W, Zheng K, Kuang H, Li Z, Wang J, Mei J. The potential of articaine as new generation of local anesthesia in dental clinics: A review. Medicine. 2022 Dec 2:101(48):e32089. doi: 10.1097/MD.0000000000032089. Epub [PubMed PMID: 36482533]
Snoeck M. Articaine: a review of its use for local and regional anesthesia. Local and regional anesthesia. 2012:5():23-33. doi: 10.2147/LRA.S16682. Epub 2012 Jun 5 [PubMed PMID: 22915899]
Su N, Liu Y, Yang X, Shi Z, Huang Y. Efficacy and safety of mepivacaine compared with lidocaine in local anaesthesia in dentistry: a meta-analysis of randomised controlled trials. International dental journal. 2014 Apr:64(2):96-107. doi: 10.1111/idj.12087. Epub 2014 Jan 16 [PubMed PMID: 24428507]
Level 1 (high-level) evidenceBrockmann WG. Mepivacaine: a closer look at its properties and current utility. General dentistry. 2014 Nov-Dec:62(6):70-5; quiz 76 [PubMed PMID: 25369391]
Su N, Wang H, Zhang S, Liao S, Yang S, Huang Y. Efficacy and safety of bupivacaine versus lidocaine in dental treatments: a meta-analysis of randomised controlled trials. International dental journal. 2014 Feb:64(1):34-45. doi: 10.1111/idj.12060. Epub 2013 Oct 11 [PubMed PMID: 24117122]
Level 1 (high-level) evidenceBecker DE, Reed KL. Essentials of local anesthetic pharmacology. Anesthesia progress. 2006 Fall:53(3):98-108; quiz 109-10 [PubMed PMID: 17175824]
Lee HS. Recent advances in topical anesthesia. Journal of dental anesthesia and pain medicine. 2016 Dec:16(4):237-244. doi: 10.17245/jdapm.2016.16.4.237. Epub 2016 Dec 31 [PubMed PMID: 28879311]
Level 3 (low-level) evidenceMathison M, Pepper T. Local Anesthesia Techniques in Dentistry and Oral Surgery. StatPearls. 2025 Jan:(): [PubMed PMID: 35593805]
Dempsy Chengappa MM, Prashanth AK. Evaluation of efficacy of computer-controlled local anaesthetic delivery system vs traditional injection system for minor pediatric surgical procedures in children. Medical journal, Armed Forces India. 2022 Sep:78(Suppl 1):S89-S95. doi: 10.1016/j.mjafi.2020.08.010. Epub 2020 Nov 2 [PubMed PMID: 36147427]
Janik K, Niemczyk W, Peterek R, Rój R, Balicz A, Morawiec T. Computer-Controlled Local Anaesthesia Delivery efficacy - a literature review. The Saudi dental journal. 2024 Aug:36(8):1066-1071. doi: 10.1016/j.sdentj.2024.05.012. Epub 2024 May 31 [PubMed PMID: 39176166]