Definition/Introduction
Overview
Coronary perfusion pressure (CPP) is the pressure gradient that drives blood flow through the coronary arteries, ensuring myocardial oxygen delivery. CPP is calculated by subtracting left ventricular end-diastolic pressure (LVEDP) from aortic diastolic pressure (ADP), as follows:
CPP = ADP – LVEDP
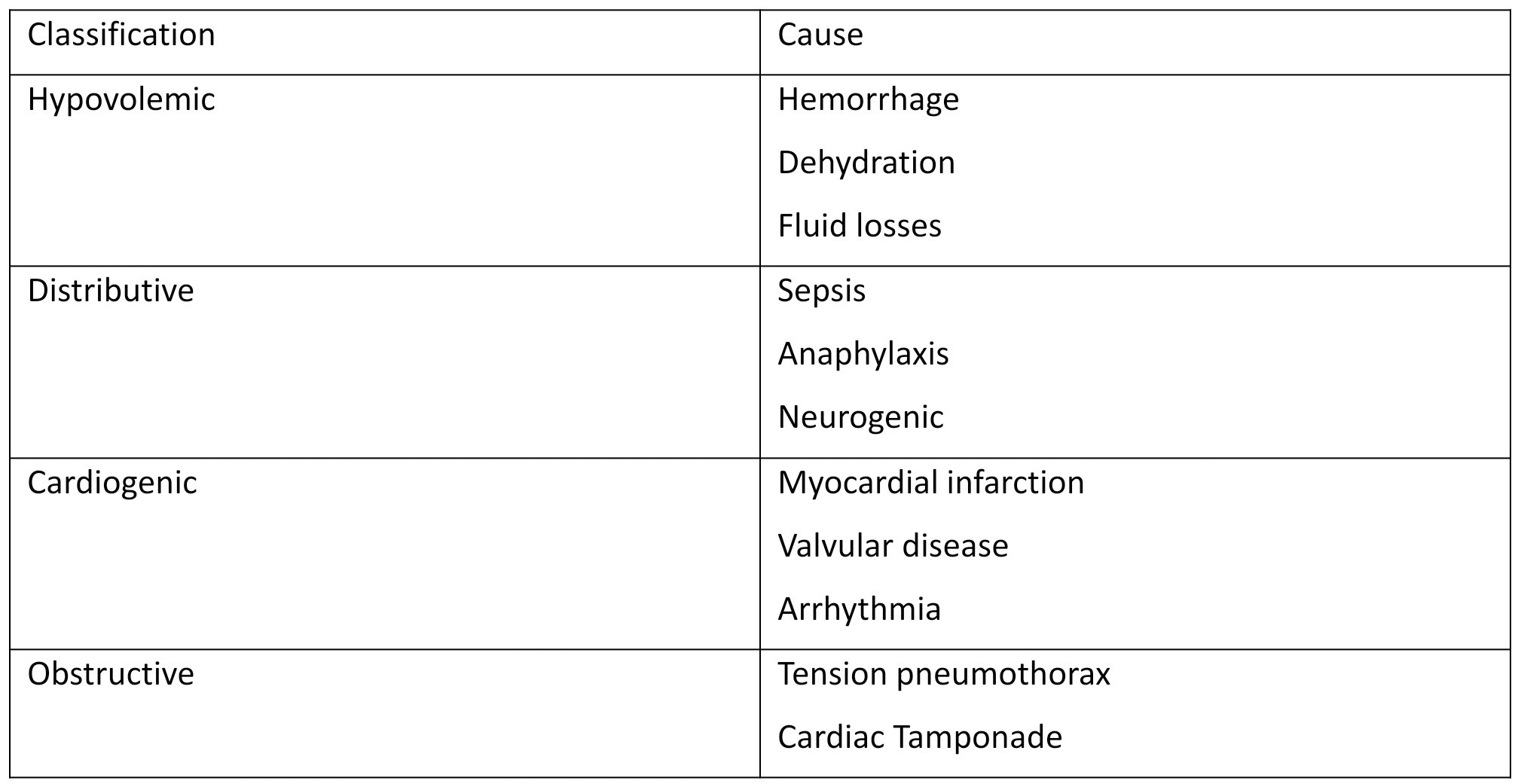
CPP is influenced by physiological alterations in hemodynamics, such as hypotension from any cause and increased LVEDP from acute heart failure. Monitoring and maintaining the required CPP are clinically crucial in patients experiencing shock or undergoing interventions to ensure myocardial viability (see Image. Causes and Classification of Shock).
Myocardial oxygen extraction is the highest of any organ at approximately 70% to 80% under resting conditions, leaving little reserve. CPP impairment can lead to ischemia, myocardial dysfunction, and even irreversible damage, particularly in the presence of stress or cardiovascular diseases. Adequate CPP ensures sufficient oxygen delivery, especially when demand increases, such as during exercise or a tachycardic state. Patients with acute coronary syndrome (ACS) or heart failure must maintain adequate CPP to sustain myocardial function during acute stress. Prolonged reductions in CPP can cause myocardial stunning, infarction, or cardiogenic shock, creating a cycle that further reduces CPP. Preserving the CPP is also vital in ACS to limit infarct size and ensure perfusion to viable myocardium.[1][2]
CPP during cardiopulmonary resuscitation (CPR) correlates with myocardial blood flow and the return of spontaneous circulation (ROSC). CPP measurement may only be performed in intensive care settings where intraarterial access can be established. A value of at least 15 mm Hg predicts the return of spontaneous circulation.
Physiology
The difference between ADP and LVEDP determines CPP in the left ventricle.[3] CPP depends on diastolic pressures because left ventricular myocardial perfusion occurs during diastole rather than systole. Both the right and left coronary arteries originate from the coronary sinuses at the aortic root before branching into the right coronary and left circumflex and anterior descending arteries.[4] Therefore, the aortic root provides the driving pressure for coronary blood flow. These arteries course along the epicardial surface before penetrating the myocardium to form subendocardial plexuses that perfuse the myocardium.
Myocardial contraction during systole compresses the arterial branches within the myocardium, preventing perfusion. Consequently, coronary perfusion occurs during diastole. LVEDP is subtracted from ADP because coronary blood flow proceeds from the epicardial to the endocardial regions.
While high left ventricular pressures are required to drive systemic circulation, the right ventricle generates lower pressures to sustain pulmonary circulation. Therefore, right ventricular pressures are far lower than the pressures exerted by the left ventricle. Right ventricular perfusion occurs predominantly in systole because systolic aortic pressure exceeds systolic right ventricular pressure. The right ventricle is also perfused to a lesser extent during diastole, driven by the smaller pressure gradient between the ADP and the right ventricular end-diastolic pressure.[5]
Technique
CPP measurement requires arterial access, typically through the femoral or radial artery. An aortic pressure catheter records the ADP, while a left ventricular pressure catheter, such as a pigtail catheter, measures LVEDP. For example, if the ADP is 80 mm Hg and the LVEDP is 20 mm Hg, the CPP is calculated as 80 - 20 = 60 mm Hg. In cardiogenic shock or critical illness, continuous monitoring of aortic and ventricular pressures offers dynamic insights into CPP variations during interventions.
This activity aims to achieve the following:
- Define CPP and identify the pressures from which it is derived.
- Describe the CPP's contribution to coronary blood flow.
- Explain CPP alterations associated with cardiac disease.
- Discuss the role of CPP reduction in type 2 myocardial infarction.
- Explore therapeutic strategies for modifying CPP in cardiovascular disease.
Issues of Concern
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Issues of Concern
Challenges in Measuring Coronary Perfusion Pressure
Measuring CPP requires invasive catheter-based techniques, which involve risks such as bleeding, infection, and arrhythmias. Factors such as improper catheter positioning, calibration errors, and arrhythmias can also compromise measurement accuracy.
Physiological Factors Affecting Coronary Perfusion Pressure
CPP varies with changes in preload, afterload, heart rate, and contractility. Tachycardia shortens the diastole and reduces CPP.[6] Conditions like heart failure and hypertrophic cardiomyopathy elevate LVEDP, diminishing the effective CPP.
Limitations of Therapeutic Strategies
Vasopressors can elevate ADP in hypotensive patients but may also increase myocardial oxygen demand, exacerbating ischemia. Excessive vasoconstriction can further impair coronary flow. Devices such as intraaortic balloon pumps (IABPs) or Impella may not consistently enhance CPP in cases of severe cardiac dysfunction.
CPP is not the sole determinant of coronary blood flow. While CPP drives coronary perfusion, coronary autoregulation ensures coronary blood flow matches myocardial demand across a CPP range of 60 to 180 mm Hg.[7] CPP represents the pressure gradient across the coronary vasculature, while autoregulation adjusts vascular resistance to maintain appropriate flow rates.
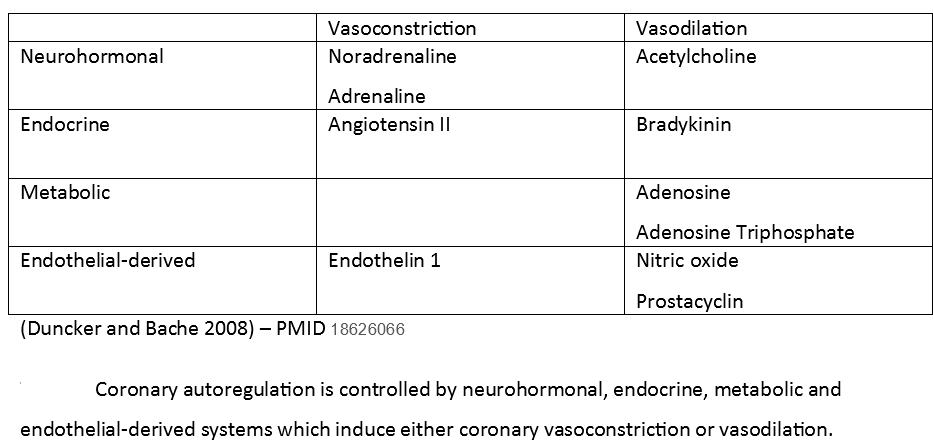
Multiple factors influence coronary vasoconstriction and vasodilation during autoregulation, categorized as neurohormonal, endocrine, metabolic, and endothelial-derived (see Image. Coronary Autoregulatory Factors). For example, systemic hypoxia and reduced coronary perfusion can lead to myocardial ischemia. Increasing coronary blood flow through elevated CPP and coronary vasodilation are the primary mechanisms for enhancing myocardial oxygen delivery under such circumstances.[8]
Coronary Perfusion Pressure and Fractional Flow Reserve
CPP assesses global coronary perfusion, while fractional flow reserve (FFR) evaluates the localized effects of stenosis. FFR is the ratio of distal-to-proximal pressure across a coronary lesion. CPP informs interventions, such as hemodynamic support with IABPs or vasopressors, whereas fractional flow reserve guides revascularization strategies, including percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG).[9][10]
Clinical Significance
Coronary Perfusion Pressure and Heart Failure
CPP is reduced in common cardiac conditions, including heart failure, making affected individuals more susceptible to myocardial ischemia. LVEDP increases in systolic heart failure, reducing CPP and left ventricular perfusion.[11] LVEDP also rises in diastolic heart failure.[12] Compensatory increases in sympathetic drive initially enhance myocardial contractility and elevate blood pressure, increasing ADP to sustain systemic and coronary blood flow. However, higher systolic blood pressure also raises cardiac afterload and promotes cardiac remodeling. Consequently, myocardial oxygen demand rises due to myocardial hypertrophy and elevated afterload against a backdrop of raised LVEDP, further heightening the myocardium’s vulnerability to ischemia.[13]
Coronary Perfusion Pressure and Coronary Artery Disease
Atherosclerotic plaques restrict blood flow through the coronary circulation, requiring compensatory coronary vasodilation distal to the plaque to maintain coronary flow and myocardial oxygen delivery. As stenosis progresses, coronary flow becomes increasingly dependent on CPP. Myocardial ischemia occurs when CPP cannot sustain coronary perfusion due to the failure of autoregulation.[14][15]
Coronary Perfusion Pressure and Myocardial Infarction
Various factors influence CPP in myocardial infarction. Hypotension due to shock, arrhythmias, or excessive vasodilation can lower ADP and reduce CPP. Acute left ventricular dysfunction resulting from ischemia or infarction can increase LVEDP, further decreasing CPP. Additionally, tachycardia arising from myocardial infarction shortens diastole, reducing CPP.
Early revascularization strategies, such as PCI and thrombolysis, are essential for restoring coronary blood flow and improving CPP. In severe cases, tailored therapies like IABPs and Impella devices can be life-saving, especially when CPP is critically low. Mean arterial pressure (MAP) should be maintained at or greater than 65 to 70 mm Hg. Vasopressors such as norepinephrine can help maintain ADP if hypotension is present.[16] Diuretics or vasodilators like nitroglycerin should be used cautiously to reduce preload without causing hypotension.
Glyceryl trinitrate is used in the acute management of type 1 myocardial infarction. Studies have shown that low-dose glyceryl trinitrate administration reduces LVEDP without lowering ADP, thereby increasing CPP.[17] The primary action of glyceryl trinitrate is central venous dilation, which decreases cardiac preload. This reduction in preload leads to a decrease in stroke volume according to the Frank-Starling law, ultimately lowering myocardial oxygen demand.[18]
The IABP is the most commonly used form of mechanical support for an acutely failing heart. The device is inserted percutaneously and positioned in the descending aorta, distal to the aortic arch. Inflation occurs during diastole, raising ADP, increasing CPP, and enhancing myocardial oxygen delivery. Additionally, the IABP reduces LVEDP and cardiac afterload, further decreasing myocardial oxygen demand. Thus, IABPs simultaneously increase myocardial oxygen supply and decrease oxygen demand.[19]
Coronary Perfusion Pressure and Type 2 Myocardial Infarction
Type 1 myocardial infarction involves the rupture of a coronary atherosclerotic plaque, leading to thrombus formation and stenosis of the arterial lumen. In contrast, type 2 myocardial infarction does not result from plaque rupture but from an imbalance between myocardial oxygen supply and demand. Decreased myocardial oxygen delivery can occur due to hypotension and reduced CPP, systemic hypoxia, or anemia. Increased myocardial oxygen demand may be caused by elevated afterload or tachyarrhythmias.
Type 2 myocardial infarction can be multifactorial. For instance, tachyarrhythmias can raise oxygen demand and reduce stroke volume, leading to hypotension and CPP reduction. Patients with preexisting cardiac conditions, such as coronary artery disease or myocardial hypertrophy, rely on CPP and are less tolerant of reductions in CPP and myocardial oxygen delivery.[20] Types 1 and 2 myocardial infarctions have similar mortality rates.[21]
Pearls
Key points to remember about CPP's clinical significance include the following:
- Left ventricular myocardial perfusion primarily occurs during diastole as a consequence of coronary anatomy.
- CPP is a significant determinant of myocardial oxygen supply, with local regulatory mechanisms maintaining coronary flow across a range of pressures.
- Acute CPP reduction during circulatory shock can lead to type 2 myocardial infarction, which has a distinct etiology compared to type 1 myocardial infarction.
Recognizing the significance of CPP helps distinguish between myocardial infarction types and manage patient outcomes more effectively.
Nursing, Allied Health, and Interprofessional Team Interventions
Understanding CPP physiology is crucial to effectively managing reductions in this parameter. Physicians and nurses must be familiar with the hemodynamic factors that influence CPP and know how to address abnormalities when they occur.
CPP is the pressure gradient that drives blood flow to the coronary arteries during CPR. High-quality chest compressions and appropriate adjunctive therapies are critical to maintaining adequate CPP. The code team leader directs interventions for optimizing hemodynamics, including medication administration, airway management, and advanced procedures.[22] Cardiologists contribute by identifying reversible causes of CPP reduction, such as coronary ischemia or tamponade, and supporting advanced mechanical circulatory interventions for sustaining adequate CPP, including extracorporeal membrane oxygenation.
Critical care nurses play a vital role in managing intravenous access, ensuring timely administration of vasopressors, and facilitating the transition to advanced resuscitative therapies. Effective strategies for improving CPP during CPR include delivering high-quality CPR, administering vasopressors, and maintaining a low ventilation rate to minimize intrathoracic pressures, enhance venous return, and support CPP. Research has demonstrated that higher CPP correlates with improved survival rates, emphasizing the importance of protocolized, high-quality CPR delivered by an interprofessional team.[23][24][25]
Nursing, Allied Health, and Interprofessional Team Monitoring
Frequent monitoring of vital signs is essential to detect changes in a patient's condition that may necessitate adjustments in clinical management. An interprofessional team approach promotes coordinated and effective management to optimize CPP during life-threatening situations. Clear and efficient communication among team members is vital during CPR to sustain CPP. The collaborative efforts of the healthcare team aim to promptly identify reversible causes (ie, the H's and T's). Advanced CPP monitoring includes invasive arterial monitoring (if time permits) to directly measure ADP and the use of end-tidal carbon dioxide as an indirect marker for perfusion quality. An end-tidal carbon dioxide of at least 10 to 20 mm Hg suggests adequate chest compressions and perfusion.
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
References
Duncker DJ, Bache RJ. Regulation of coronary blood flow during exercise. Physiological reviews. 2008 Jul:88(3):1009-86. doi: 10.1152/physrev.00045.2006. Epub [PubMed PMID: 18626066]
Level 3 (low-level) evidenceFeigl EO. Coronary physiology. Physiological reviews. 1983 Jan:63(1):1-205 [PubMed PMID: 6296890]
Nguyen T, Do H, Pham T, Vu LT, Zuin M, Rigatelli G. Left ventricular dysfunction causing ischemia in patients with patent coronary arteries. Perfusion. 2018 Mar:33(2):115-122. doi: 10.1177/0267659117727826. Epub 2017 Aug 21 [PubMed PMID: 28823216]
Loukas M, Sharma A, Blaak C, Sorenson E, Mian A. The clinical anatomy of the coronary arteries. Journal of cardiovascular translational research. 2013 Apr:6(2):197-207. doi: 10.1007/s12265-013-9452-5. Epub 2013 Feb 20 [PubMed PMID: 23423864]
Level 3 (low-level) evidenceBrooks H, Kirk ES, Vokonas PS, Urschel CW, Sonnenblick EH. Performance of the right ventricle under stress: relation to right coronary flow. The Journal of clinical investigation. 1971 Oct:50(10):2176-83 [PubMed PMID: 5116207]
Level 3 (low-level) evidenceHeusch G. Heart rate in the pathophysiology of coronary blood flow and myocardial ischaemia: benefit from selective bradycardic agents. British journal of pharmacology. 2008 Apr:153(8):1589-601. doi: 10.1038/sj.bjp.0707673. Epub 2008 Jan 28 [PubMed PMID: 18223669]
Level 3 (low-level) evidenceFeigl EO. Coronary autoregulation. Journal of hypertension. Supplement : official journal of the International Society of Hypertension. 1989 Sep:7(4):S55-8; discussion S59 [PubMed PMID: 2681597]
Level 3 (low-level) evidenceGoodwill AG, Dick GM, Kiel AM, Tune JD. Regulation of Coronary Blood Flow. Comprehensive Physiology. 2017 Mar 16:7(2):321-382. doi: 10.1002/cphy.c160016. Epub 2017 Mar 16 [PubMed PMID: 28333376]
Foley MJ, Rajkumar CA, Ahmed-Jushuf F, Simader F, Chotai S, Seligman H, Macierzanka K, Davies JR, Keeble TR, O'Kane P, Haworth P, Routledge H, Kotecha T, Clesham G, Williams R, Din J, Nijjer SS, Curzen N, Sinha M, Petraco R, Spratt J, Sen S, Cole GD, Harrell FE Jr, Howard JP, Francis DP, Shun-Shin MJ, Al-Lamee R, ORBITA-2 investigators. Fractional Flow Reserve and Instantaneous Wave-Free Ratio as Predictors of the Placebo-Controlled Response to Percutaneous Coronary Intervention in Stable Coronary Artery Disease. Circulation. 2024 Oct 27:():. doi: 10.1161/CIRCULATIONAHA.124.072281. Epub 2024 Oct 27 [PubMed PMID: 39462291]
Lüscher TF. Chronic coronary syndromes: perfusion pressure, FFR, and secondary prevention. European heart journal. 2020 May 1:41(17):1611-1614. doi: 10.1093/eurheartj/ehaa380. Epub [PubMed PMID: 33216867]
Banerjee P. Heart failure: a story of damage, fatigue and injury? Open heart. 2017:4(2):e000684. doi: 10.1136/openhrt-2017-000684. Epub 2017 Oct 15 [PubMed PMID: 29081980]
Choi S, Shin JH, Park WC, Kim SG, Shin J, Lim YH, Lee Y. Two Distinct Responses of Left Ventricular End-Diastolic Pressure to Leg-Raise Exercise in Euvolemic Patients with Exertional Dyspnea. Korean circulation journal. 2016 May:46(3):350-64. doi: 10.4070/kcj.2016.46.3.350. Epub 2016 May 27 [PubMed PMID: 27275172]
Zhang DY, Anderson AS. The sympathetic nervous system and heart failure. Cardiology clinics. 2014 Feb:32(1):33-45, vii. doi: 10.1016/j.ccl.2013.09.010. Epub [PubMed PMID: 24286577]
Cruickshank JM. Clinical importance of coronary perfusion pressure in the hypertensive patient with left ventricular hypertrophy. Cardiology. 1992:81(4-5):283-90 [PubMed PMID: 1301256]
Level 3 (low-level) evidenceMahendiran T, Collet C, De Bruyne B. Coronary-Artery Autoregulation with Increasing Stenosis. The New England journal of medicine. 2024 Jun 6:390(21):2030-2032. doi: 10.1056/NEJMc2402216. Epub [PubMed PMID: 38838318]
Bloom JE, Chan W, Kaye DM, Stub D. State of Shock: Contemporary Vasopressor and Inotrope Use in Cardiogenic Shock. Journal of the American Heart Association. 2023 Aug:12(15):e029787. doi: 10.1161/JAHA.123.029787. Epub 2023 Jul 25 [PubMed PMID: 37489740]
Yaginuma T, Avolio A, O'Rourke M, Nichols W, Morgan JJ, Roy P, Baron D, Branson J, Feneley M. Effect of glyceryl trinitrate on peripheral arteries alters left ventricular hydraulic load in man. Cardiovascular research. 1986 Feb:20(2):153-60 [PubMed PMID: 3085950]
Sequeira V, van der Velden J. Historical perspective on heart function: the Frank-Starling Law. Biophysical reviews. 2015 Dec:7(4):421-447. doi: 10.1007/s12551-015-0184-4. Epub 2015 Nov 19 [PubMed PMID: 28510104]
Level 3 (low-level) evidenceParissis H, Graham V, Lampridis S, Lau M, Hooks G, Mhandu PC. IABP: history-evolution-pathophysiology-indications: what we need to know. Journal of cardiothoracic surgery. 2016 Aug 4:11(1):122. doi: 10.1186/s13019-016-0513-0. Epub 2016 Aug 4 [PubMed PMID: 27487772]
Gupta S, Vaidya SR, Arora S, Bahekar A, Devarapally SR. Type 2 versus type 1 myocardial infarction: a comparison of clinical characteristics and outcomes with a meta-analysis of observational studies. Cardiovascular diagnosis and therapy. 2017 Aug:7(4):348-358. doi: 10.21037/cdt.2017.03.21. Epub [PubMed PMID: 28890871]
Level 1 (high-level) evidenceChapman AR, Adamson PD, Mills NL. Assessment and classification of patients with myocardial injury and infarction in clinical practice. Heart (British Cardiac Society). 2017 Jan 1:103(1):10-18. doi: 10.1136/heartjnl-2016-309530. Epub 2016 Nov 2 [PubMed PMID: 27806987]
Bennett CE. Not Who, but Rather How: The Ideal Resuscitation Team Leader. Mayo Clinic proceedings. Innovations, quality & outcomes. 2021 Oct:5(5):817-819. doi: 10.1016/j.mayocpiqo.2021.07.004. Epub 2021 Aug 17 [PubMed PMID: 34458679]
Level 2 (mid-level) evidenceJohansson S, Rosengren A, Young K, Jennings E. Mortality and morbidity trends after the first year in survivors of acute myocardial infarction: a systematic review. BMC cardiovascular disorders. 2017 Feb 7:17(1):53. doi: 10.1186/s12872-017-0482-9. Epub 2017 Feb 7 [PubMed PMID: 28173750]
Level 1 (high-level) evidenceSutton RM, Friess SH, Maltese MR, Naim MY, Bratinov G, Weiland TR, Garuccio M, Bhalala U, Nadkarni VM, Becker LB, Berg RA. Hemodynamic-directed cardiopulmonary resuscitation during in-hospital cardiac arrest. Resuscitation. 2014 Aug:85(8):983-6. doi: 10.1016/j.resuscitation.2014.04.015. Epub 2014 Apr 28 [PubMed PMID: 24783998]
Level 3 (low-level) evidenceParadis NA, Martin GB, Rivers EP, Goetting MG, Appleton TJ, Feingold M, Nowak RM. Coronary perfusion pressure and the return of spontaneous circulation in human cardiopulmonary resuscitation. JAMA. 1990 Feb 23:263(8):1106-13 [PubMed PMID: 2386557]