Introduction
Premature ventricular complexes (PVCs), or premature ventricular contractions, are ectopic beats that arise from within the ventricles. Also known as premature ventricular contractions, the American Heart Association (AHA) and Heart Rhythm Society prefer the term "complexes" as electric activity does not always result in cardiac muscle contraction.[1] They are common and can occur in various clinical scenarios and diverse populations. They can present in patients both with and without preexisting cardiac disease.[2] Reports show that frequent PVCs can cause progressive left ventricular (LV) dysfunction and dilation, which may return to normal after successful catheter ablation of the PVCs.[3][4]
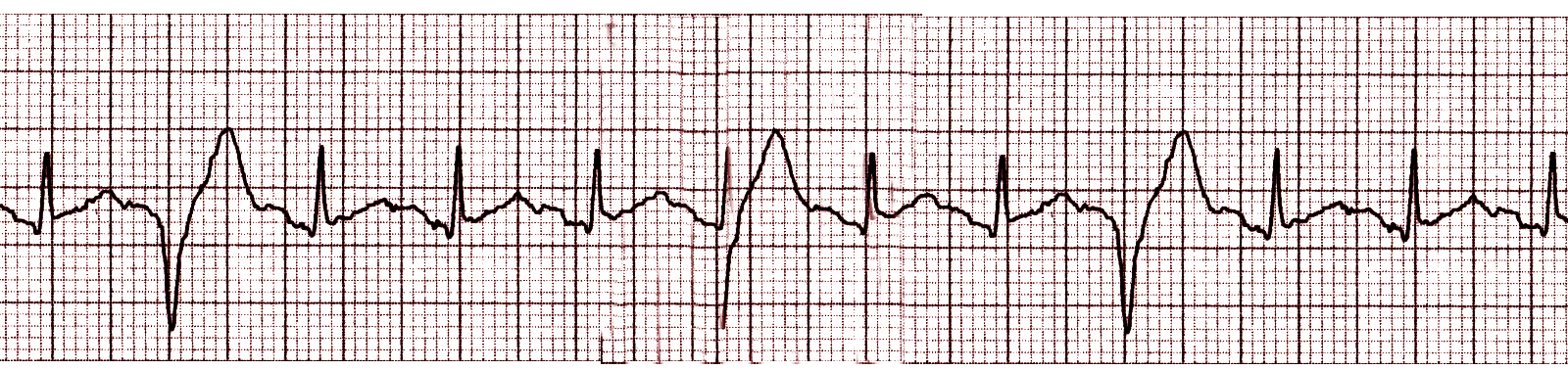
The morphology of PVCs is highly variable, and it depends upon their place of origin, underlying structural disease, or the use of antiarrhythmic drugs. PVCs usually originate from the ventricle, leading to a prolonged QRS complex duration greater than 120 ms because of the delay in the spread of activation to the contralateral ventricle through the nonspecialized myocardium. However, in certain instances, a QRS complex of duration less than 120 milliseconds can occur, leading to activation of both ventricles "synchronously" because of electrical stimulus from one of the fascicles through a specific conduction system (see Image. Premature Ventricular Complexes).[5]
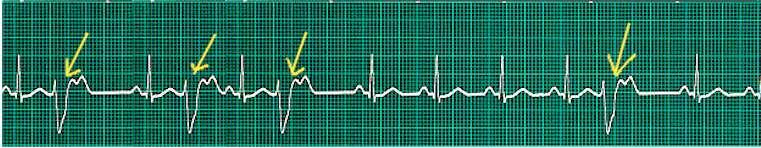
PVCs are classified according to coupling interval (early and delayed), QRS duration (wide and narrow), morphology, and complexity. The 12-lead electrocardiogram (ECG) helps identify the precise location of the origin of PVCs only by the morphology of ectopic beats (see Image. Premature Ventricular Complex on Electrocardiogram). PVCs originating in the left ventricle are usually associated with a right bundle branch block (RBBB), and those that arise in the right ventricle show a left bundle branch block (LBBB) pattern.
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Identifying the etiological cause requires a thorough history and physical examination. While most PVCs occur spontaneously without an identifiable cause, several known etiologies exist. PVCs can arise from numerous risk factors or causes, with some occurring benignly due to excessive adrenergic stimulation. PVCs stem from a broad range of cardiac and noncardiac conditions, lifestyle factors, and demographic risks. While some PVCs are benign and self-limited, understanding their etiology is vital for appropriate evaluation and management. Common known etiologies include excess caffeine consumption, excess catecholamines,[6] high levels of anxiety, and electrolyte abnormalities.
Noncardiac conditions like hypercapnia, hyperthyroidism, anemia, and sleep deprivation can provoke PVCs, as can acute cardiac events, eg, myocardial infarction, myocarditis, myocardial contusion, mitral valve prolapse, and cardiomyopathies (particularly hypertrophic and dilated forms). Structural heart diseases that disrupt conduction pathways are frequent culprits. Other factors include various medications, electrolyte abnormalities, and hypoxia. The following describes several etiologies of PVCs:
- Illicit drug abuse (eg, alcohol, amphetamines, and cocaine)
- Various medications eg, digoxin, tricyclic antidepressants, sympathomimetics, aminophylline, caffeine
- Electrolyte imbalance, including hypokalemia, hypomagnesemia, or hypercalcemia
- Hypoxia
- Hypercapnia
- Acute myocardial infarction
- Cardiomyopathies mainly hypertrophic and dilated cardiomyopathy
- Myocarditis
- Myocardial contusion
- Mitral valve prolapse [6]
Risk factors for Premature Ventricular Complexes
Specific populations and demographic factors exhibit a higher risk for PVCs, including:
- Advanced age
- Male gender
- Hypertension
- African American
- hypomagnesemia
- Bundle branch block
- Hypokalemia
- Underlying ischemic heart disease [7]
Epidemiology
Studies on the prevalence of PVCs often derive from ECG or Holter monitor databases, where higher rates are expected due to the clinical setting. Early research showed a prevalence of 7.8 per 1000 over a 48-second ECG, with rates increasing with age. More recent studies, eg, the Cardiovascular Health Study, found PVCs in 1.8% to 5.2% of participants using 10-second ECGs, with prevalence rising with more extended monitoring. A 24-hour Holter monitoring detected PVCs in 69% of healthy adults in 1 study and nearly all elderly participants in another. Longer monitoring and newer wearable technologies reveal even higher detection rates, emphasizing the increasing prevalence of PVCs with age and extended observation durations.[1]
According to the Framingham Heart Study, the prevalence of PVCs was found to be higher in 1-hour continuous ECG monitoring, with 33% being men and 32% women with no history of any coronary artery disease. For those patients with coronary artery disease, the prevalence was comparatively higher, with 58% in men and women 49%.[8] The variability in the reported data differs due to the different monitoring techniques and duration used to calculate PVC prevalence. Several factors are involved in this diversity, including coronary heart disease, African-American ethnicity, male gender, older age, hypertension, faster sinus rates, lower serum potassium or magnesium levels, and lower education level.[9]
Pathophysiology
The pathophysiological mechanisms for underlying PVCs are often unclear, with multiple processes potentially contributing. The primary mechanisms thought to result in PVCs include triggered activity, automaticity, and reentry. Identification of the pathophysiological is essential as this informs the management approach.[1]
Triggered activity arises during the early depolarization phase due to exceeding the cell membrane activation threshold. The PVCs associated with bradycardia are often caused by this mechanism, along with other factors like electrolyte abnormalities, ischemia, and digoxin toxicity. Caffeine and catecholamines may exacerbate this mechanism, while β-blockers, adenosine, and calcium channel blockers can reduce its effects.[1][10][1]
Automaticity involves PVCs originating from abnormal cardiomyocyte activity, often showing parasystole. The enhanced automaticity characteristically results from an ectopic ventricular focus that fires spontaneously. A variety of stimuli, including myocardial ischemia, electrolyte imbalance, and heightened adrenergic state, can cause enhanced automaticity.[1]
Though usually linked to sustained arrhythmias, reentry can also cause PVCs. It requires 2 distinct pathways with unidirectional block, often involving scars, fibrosis, or abnormal conduction in specific regions. This mechanism is seen in phenomena like bundle-branch or fascicular PVCs.[1]
History and Physical
Clinical Features of Premature Ventricular Complexes
Most often, PVCs are asymptomatic. However, some patients report fatigue, palpitations, lightheadedness, or dizziness. In patients with preexisting heart disease, frequent PVCs can cause dyspnea, angina, and palpitations. Patients may often report a sense of abnormal neck pulsation with palpitations when they experience PVCs. PVCs rarely cause hemodynamic compromise, leading to presyncope or syncope, mainly if sustained or frequent runs occur. In asymptomatic individuals, PVCs may be initially identified on a routine physical exam by an irregular rhythm consisting of a premature beat followed by a compensatory pause with subsequent return of regular rhythm or cardiac auscultation revealing a sharp first heart sound or splitting of S2 or on a routine office electrocardiogram.
In some cases, PVCs are the first manifestation of underlying structural heart disease. For this reason, in all patients with PVCs, one must perform a detailed clinical history of 12-lead EKG and chest x-ray. The indication for further evaluation by Holter monitoring, stress test, or echocardiogram depends on whether PVCs persist after the initial assessment of suspected structural heart disease or the onset of complex forms of ventricular arrhythmias.
For over 20 years, the accepted view is that the presence of PVCs in the absence of structural heart disease presents a favorable, benign prognosis, even when they arise frequently. Therefore, treatment strategies for PVCs are planned according to the frequency of occurrence and symptoms. In some cases, very frequent PVCs can produce cardiomyopathy, being an indication for treatment, even in the absence of symptoms. However, the clinical importance of frequent PVCs in patients without any left ventricular dysfunction is still unclear.
Evaluation
Electrocardiograph Evaluation
The initial workup should start with a resting ECG. The PVC is electrocardiographically defined as a premature QRS complex with an abnormal morphology and duration greater than 120 milliseconds. A large T-wave classically follows it with discordant deflection (opposite polarity) from the QRS complex. Typically, no preceding P waves are present; however, the impulse generated from the PVCs can often be propagated in a retrograde fashion to the atria and can cause a classic compensatory pause following the PVC. This pause is due to the atrioventricular (AV) nodal blockade due to depolarization from the retrograde impulse and subsequent refractory period of the AV node. Though the sinus node fires at the appropriate time, its impulse is not propagated, as the AV node has been rendered refractory, thus causing an R-R interval that is precisely double the intrinsic R-R interval.
Usually, a PVC is followed by a full compensatory pause because the sinus node timing is not interrupted; 1 sinus P-wave is not able to reach the ventricles because they are still refractory from the PVC; the following sinus impulse thus occurs on time based on the sinus rate. In contrast, premature atrial contractions (PACs) are usually associated with an incomplete pause because the PAC usually enters the sinoatrial node and resets its timing, which enables the following sinus impulse to appear earlier than expected. Not all PVCs, however, get followed by a pause. If a PVC occurs early enough, especially with prolonged heart rates, it may appear sandwiched between the 2 normal heartbeats. This phenomenon is called an "interpolated PVC." The sinus impulse following the PVC is conducted with a longer PR interval because of the retrograde concealed conduction by the premature ventricular impulse into the AV-node junction, slowing the subsequent conduction of the sinus impulse.
The morphology of the QRS complex can be a means to determine the origin of the PVC. Left and right ventricular outflow tracts are the most common sites of origin of PVCs and account for approximately two-thirds of all PVCs. The electrocardiographic morphology can similarly determine the ectopic origin of the source of ventricular tachycardia. PVCs with the morphology of a typical left bundle branch block (LBBB) and an inferior axis originate from the right ventricular outflow tract, while PVCs with the morphology of a standard right bundle branch block (RBBB) and an inferior axis originate from the left ventricular outflow tract. PVCs can originate from numerous other locations, eg, the ventricular free wall, ventricular septum, aortic cusp, tricuspid or mitral annuli, pulmonary artery, fascicles, or papillary muscles.
Individuals with multiple abnormal QRS complexes are classified as having multifocal PVCs, which are more common than unifocal PVCs. PVCs are also definable by the pattern in which they occur relative to intrinsic beats. Bigeminy indicates a PVC following a single, standard QRS complex, whereas trigeminy indicates a PVC following 2 normal QRS complexes. Multiple successive PVCs also have a distinct annotation; 2 PVCs successively a couplet, 3 PVCs successively a triplet, and more than 3 successive PVCs classify as a run of PVCs or ventricular tachycardia.
When more than 3 consecutive PVCs are present, the term used is nonsustained ventricular tachycardia, and the term sustained ventricular tachycardia is used when the duration lasts >30 seconds. Couplets can be further defined by the interval between the ectopic PVCs and the subsequent usually conducted beat from the sinoatrial node. A stable interval between the PVCs and a regular beat is called fixed coupling. Variable coupling is when these intervals change from beat to beat. PVCs are considered "frequent" if they occur >30 times per hour or >20% of total heartbeats.
In addition to a resting ECG, the clinician should obtain ambulatory ECG monitoring for a minimum of 24 hours with a device such as a Holter monitor. If patients are particularly symptomatic, and the initial 24-hour monitoring is unrevealing, an event recorder or loop recorder can provide prolonged monitoring. In patients with PVCs, the ECG may reveal other findings that include:
- Electrolyte abnormalities (peaked T waves, QT prolongation)
- Left ventricular hypertrophy
- With an old MI, one may see Q waves, loss of R waves, and a bundle branch block
- Acute ischemia may present with ST-segment elevation/depression and/or T-wave inversion
Additional Diagnostic Studies
Evaluating for an underlying electrolyte abnormality, with specific attention to serum potassium and magnesium levels, is also reasonable. Additional laboratory evaluation with urine drug screen, thyroid stimulating hormone level, cardiac biomarkers (troponin), and serum digoxin level merit consideration when indicated. In most patients, especially those who are symptomatic, evaluation with a transthoracic echocardiogram should be considered to evaluate for underlying structural cardiac abnormalities. Further evaluation with exercise/chemical stress testing, nuclear myocardial perfusion imaging, coronary angiography, and cardiac magnetic resonance imaging (MRI) should also be considered on a case-by-case basis for each patient.
Treatment / Management
Medical Management
Patients with PVCs in the setting of underlying structural heart disease should receive guideline-directed medical therapy (GDMT) as indicated in their specific disease process. Treatment is a consideration in patients who are symptomatic with a high burden of ectopic beats or who have frequent episodes of nonsustained ventricular tachycardia. Most notably, PVCs that transform into ventricular fibrillation (usually mediated by the R-on-T phenomenon) should receive immediate defibrillation. If treatment is indicated, β-blockers are typically first-line therapy. Catecholamine-sensitive or exercise-induced PVCs have an excellent response to the initiation of β-blocker therapy. PVCs in the setting of CHF or myocardial infarction should be treated with β-blockers as first-line therapy, as directed by GDMT.
Similarly, nondihydropyridine calcium channel blockers can be a consideration in patients with a contraindication for β-blockers. Patients can start antiarrhythmic treatment with class Ic agents (eg, flecainide or propafenone) when their symptoms have not improved after initiation of β-blocker or calcium channel blocker therapy. However, these agents are proarrhythmic, increase mortality, and are contraindicated in patients with preexisting coronary artery disease. For this specific patient population, amiodarone has proven to be effective in suppressing PVCs; however, due to adverse effect profiles, the administration should generally be reserved under close clinical monitoring for patients with underlying structural heart disease or coronary disease and significant symptomatology.
Ablation Therapy
Patients who cannot tolerate the therapy should be referred to an electrophysiologist for consideration of ablation. PVC ablation is also the recommended intervention in patients who develop PVC-induced cardiomyopathy, especially in those with a significant PVC burden.[11] The definition of high PVC burden is >10,000 ectopic ventricular beats or >10% of all beats recorded on 24-hour ambulatory monitoring.
The accepted thinking is that cardiomyopathy is more likely in patients whose PVCs have a very wide QRS complex, the beats arise from the epicardium, or in patients with PVC occurring higher than a quarter of all beats on 24-hour Holter monitor (PVC burden of over 25%). This cardiomyopathy is generally considered to be reversible after ablation; however, some degree of left ventricular systolic dysfunction may rarely persist after the ablation of the ectopic focus.[12] Patients who have already developed cardiomyopathy due to high-frequency PVCs should also have a referral for radiofrequency ablation, as evidenced by a 2014 meta-analysis demonstrating that catheter ablation of PVCs originating from the right ventricular outflow tract significantly reduced ventricular ectopy burden and improved left ventricular ejection fraction. Ablation may also be indicated in patients with frequent PVCs that interfere with cardiac resynchronization therapy.[13](B3)
Several cardiology societies have provided guidelines on the utilization of ablations for patients with PVCs. The 2019 AHA, American College of Cardiology, and Heart Rhythm Society Guideline for Management of Patients with Ventricular Arrhythmia and the Prevention of Sudden Cardiac Death recommends that catheter ablation may be useful for patients with reduced left ventricular ejection fraction suspected to have been caused by frequent PVCs defined as >15% of all beats who failed to respond or could not tolerate antiarrhythmic medications.[14](B3)
The 2015 European Society of Cardiology Guidelines recommends considering ablation therapy for patients with reduced left ventricular ejection fraction and frequent symptomatic PVCs or nonsustained ventricular tachycardia.[15] Additionally, the European Heart Rhythm Association/Heart Failure Association recommends that patients with heart failure with reduced ejection fraction and a high burden of PVCs, defined as >10,000 PVCs in 24 hours, should be aggressively treated with catheter ablation if they have failed, declined, or are intolerant to antiarrhythmic therapy especially if a single dominant PVCs morphology is identified.[16][17][18][19][16](B3)
Differential Diagnosis
Differentiating PVCs from other arrhythmias can be challenging because several arrhythmias may mimic PVCs, including:
- Aberrant PACs
- Fusion beats
- Premature junctional contractions
- Idioventricular escape rhythms
- Sustained and nonsustained ventricular tachycardia
- Bigeminy, trigeminy, or quadrigeminy
- Supraventricular tachycardia
- Atrial fibrillation
- Sinus arrhythmia
Prognosis
In almost all heart diseases, due to the high incidence and complexity, PVCs can be the major cause of mortality in coronary artery disease and dilated cardiomyopathy. According to the MRIFT and Framingham Heart Study, frequent occurrence of PVCs correlates with an increased risk of sudden cardiac death.[20][21] As these studies lack the diligent measures taken to investigate underlying heart disease, they are under great criticism because they can halve the death outcome.
The benign premature ventricular contractions that occur occasionally do not interfere with the average lifespan, but their frequent occurrence implies an ominous prognosis. Patients with a previous myocardial infarction with frequent PVCs are prone to sudden cardiac death. However, patients treated either with medication or catheter ablation therapy can improve and recover from the LV dysfunction.[4]
The characteristics of PVCs that confer poor prognosis include:
- An "arrhythmic burden" with >500 PVCs in 24 hours on Holter monitoring
- PVC morphologies with LBBB or RBBB
- PVCs with intermediate or superior axis and wide QRS complexes
- Short-coupled PVCs
- PVCs superimposed on preceding T-waves
- PVCs induced by adrenergic stimulation
Healthy patients without structural heart disease who experience infrequent PVCs have the same prognosis as the general population. However, those with frequent PVCs (>1000/day) are at risk of developing dilated cardiomyopathy.[22] For those with heart disease, PVCs are an indicator of increased mortality risk.[23]
PVCs occurring during a period of exercise have a varying prognosis, depending on when they occur. If the PVCs arise during exercise, the prognosis is very good. If they occur during the recovery phase of exercise, the long-term mortality risk is higher than in the general population.[24]
Complications
Frequent PVCs are often associated with complications if remain unattended, which include:
- Left ventricular dysfunction
- Dilated cardiomyopathy
- Sustained ventricular tachycardia
- Sudden cardiac arrest
Postoperative and Rehabilitation Care
Cardiac rehabilitation is comprised of comprehensive long-term and short-term goals. The long-term goals focus on medical evaluation, cardiovascular risk assessment, health education, lifestyle modification, regular exercise motivation, counseling, and behavioral therapy. These goals alter the natural history of cardiac disease associated with PVCs, stabilize and reverse the progression of LV dysfunction, and reduce the risk of sudden cardiac death. The short-term goals consist of controlling cardiac symptoms, limiting cardiac illness's physiological and psychological effects, improving functional capacity, and boosting vocational and psychosocial status.[25][26]
Cardiac rehabilitation improves patients' outcomes by improving their quality of life, increasing their perception of physical development and motivation, altering risk factors, adjusting interpersonal roles, and potentially advancing at work.
Consultations
Patients with PVCs usually require consultation with a general physician, cardiologist, interventional cardiologist, and cardiac electrophysiologist.
Deterrence and Patient Education
Patients presenting to family physicians with symptoms like palpitations or skipped beats require detailed assessment. Although various cardiac etiologies can cause palpitations, their association with noncardiac causes like anemia, fever, drug use, or even anxiety in otherwise healthy individuals should also remain in view. Reassurance and lifestyle modification are the mainstay of management in benign occurring PVCs. The patients should also undergo screening for anxiety or panic disorder with the help of a questionnaire. Patient education about when to seek advice in case of a worsening of symptoms is essential.
With the help of an interprofessional approach, patients will have the chance to interact with multiple members of the patient care team. Relaying changes in the regimen to the patients is a vital aspect of educating the patients. Clinicians should emphasize keeping a scheduled follow-up of appointments. If warranted, patients should receive counsel and education regarding the benefits of quitting smoking and losing weight. With the help of written pamphlets or illustrations, patient education should be to enhance the patient's learning.
Pearls and Other Issues
The general population with frequent PVCs, defined as the presence of at least 1 PVC on a 12-lead ECG or >30 PVCs per hour, is usually associated with increased cardiovascular risk and mortality. The cardiac MRI and methods used to suppress the PVCs aid in reversing the LV dysfunction. Therefore, the differentiation of primary cardiomyopathy from PVC-mediated LV dysfunction is of prime importance.[27]
A detailed cardiovascular risk assessment and management is necessary for the clinical finding of PVCs on ECG of even healthy patients.[23] A poor prognosis associated with cardiac pathology caused by PVCs can be altered simply by early prevention and prompt treatment. After undergoing specific treatment or intervention, the patients should be subjected to long-term follow-up by continuous monitoring, especially in cases of arrhythmia or heart failure.[28]
Enhancing Healthcare Team Outcomes
Interprofessional collaboration is essential to optimize patient-centered care, safety, and outcomes for patients with PVCs. Effective teamwork requires healthcare professionals, including physicians, advanced practitioners, nurses, pharmacists, radiologists, and cardiac specialists, to share expertise, skills, and knowledge in a coordinated effort. The general physician often initiates the care process through thorough evaluation and timely referral to a cardiologist, who focuses on cardiovascular risk assessment, patient education, and follow-up compliance. Interventional cardiologists contribute to the diagnosis and management of ischemic heart disease and perform catheter ablation when necessary, guided by cardiac electrophysiologists who map the origin of PVCs. Radiologists support diagnosis through advanced imaging, while clinical pharmacists optimize medication management and provide education to patients and clinicians. Nurses, particularly in intensive care units or cardiology settings, are often the first to detect PVCs on monitors or ECG strips and play a critical role in patient monitoring and immediate care.
Trust, respect, and open communication among team members are vital for effective care coordination. Regular case discussions shared patient files, and interprofessional meetings enhance collaboration, ensuring that each patient's specific needs and expectations are addressed. For patients with asymptomatic PVCs and no cardiac anomalies, reassurance is often sufficient, but high-frequency PVCs or underlying conditions may require specialized intervention. In cases of persistent PVCs postmyocardial infarction, referral to an electrophysiologist is crucial. By fostering close communication and mutual respect, interprofessional teams can improve patient outcomes, reduce risks, and deliver high-quality, patient-centered care.
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
References
Marcus GM. Evaluation and Management of Premature Ventricular Complexes. Circulation. 2020 Apr 28:141(17):1404-1418. doi: 10.1161/CIRCULATIONAHA.119.042434. Epub 2020 Apr 27 [PubMed PMID: 32339046]
Wang K, Hodges M. The premature ventricular complex as a diagnostic aid. Annals of internal medicine. 1992 Nov 1:117(9):766-70 [PubMed PMID: 1384411]
Chugh SS, Shen WK, Luria DM, Smith HC. First evidence of premature ventricular complex-induced cardiomyopathy: a potentially reversible cause of heart failure. Journal of cardiovascular electrophysiology. 2000 Mar:11(3):328-9 [PubMed PMID: 10749356]
Level 3 (low-level) evidenceTakemoto M, Yoshimura H, Ohba Y, Matsumoto Y, Yamamoto U, Mohri M, Yamamoto H, Origuchi H. Radiofrequency catheter ablation of premature ventricular complexes from right ventricular outflow tract improves left ventricular dilation and clinical status in patients without structural heart disease. Journal of the American College of Cardiology. 2005 Apr 19:45(8):1259-65 [PubMed PMID: 15837259]
Shi LR. [Clinical significance of the site of origin of premature ventricular contractions (PVC)]. Zhonghua xin xue guan bing za zhi. 1983 Mar:11(1):36-8 [PubMed PMID: 6192983]
Frigy A, Csiki E, Caraşca C, Szabó IA, Moga VD. Autonomic influences related to frequent ventricular premature beats in patients without structural heart disease. Medicine. 2018 Jul:97(28):e11489. doi: 10.1097/MD.0000000000011489. Epub [PubMed PMID: 29995813]
Ribeiro WN, Yamada AT, Grupi CJ, da Silva GT, Mansur AJ. Premature atrial and ventricular complexes in outpatients referred from a primary care facility. PloS one. 2018:13(9):e0204246. doi: 10.1371/journal.pone.0204246. Epub 2018 Sep 20 [PubMed PMID: 30235300]
Morshedi-Meibodi A, Evans JC, Levy D, Larson MG, Vasan RS. Clinical correlates and prognostic significance of exercise-induced ventricular premature beats in the community: the Framingham Heart Study. Circulation. 2004 May 25:109(20):2417-22 [PubMed PMID: 15148273]
Simpson RJ Jr, Cascio WE, Schreiner PJ, Crow RS, Rautaharju PM, Heiss G. Prevalence of premature ventricular contractions in a population of African American and white men and women: the Atherosclerosis Risk in Communities (ARIC) study. American heart journal. 2002 Mar:143(3):535-40 [PubMed PMID: 11868062]
Level 2 (mid-level) evidenceKaraman K, Karayakali M, Arisoy A, Akar I, Ozturk M, Yanik A, Yilmaz S, Celik A. Is There any Relationship Between Myocardial Repolarization Parameters and the Frequency of Ventricular Premature Contractions? Arquivos brasileiros de cardiologia. 2018 Jun:110(6):534-541. doi: 10.5935/abc.20180079. Epub [PubMed PMID: 30226912]
Zhu DW, Maloney JD, Simmons TW, Nitta J, Fitzgerald DM, Trohman RG, Khoury DS, Saliba W, Belco KM, Rizo-Patron C. Radiofrequency catheter ablation for management of symptomatic ventricular ectopic activity. Journal of the American College of Cardiology. 1995 Oct:26(4):843-9 [PubMed PMID: 7560606]
Lee AK, Deyell MW. Premature ventricular contraction-induced cardiomyopathy. Current opinion in cardiology. 2016 Jan:31(1):1-10. doi: 10.1097/HCO.0000000000000236. Epub [PubMed PMID: 26599061]
Level 3 (low-level) evidenceNoheria A, Deshmukh A, Asirvatham SJ. Ablating Premature Ventricular Complexes: Justification, Techniques, and Outcomes. Methodist DeBakey cardiovascular journal. 2015 Apr-Jun:11(2):109-20. doi: 10.14797/mdcj-11-2-109. Epub [PubMed PMID: 26306129]
Cronin EM, Bogun FM, Maury P, Peichl P, Chen M, Namboodiri N, Aguinaga L, Leite LR, Al-Khatib SM, Anter E, Berruezo A, Callans DJ, Chung MK, Cuculich P, d'Avila A, Deal BJ, Della Bella P, Deneke T, Dickfeld TM, Hadid C, Haqqani HM, Neal Kay G, Latchamsetty R, Marchlinski F, Miller JM, Nogami A, Patel AR, Pathak RK, Saenz Morales LC, Santangeli P, Sapp JL Jr, Sarkozy A, Soejima K, Stevenson WG, Tedrow UB, Tzou WS, Varma N, Zeppenfeld K. 2019 HRS/EHRA/APHRS/LAHRS expert consensus statement on catheter ablation of ventricular arrhythmias. Journal of arrhythmia. 2019 Jun:35(3):323-484. doi: 10.1002/joa3.12185. Epub 2019 May 10 [PubMed PMID: 31293696]
Level 3 (low-level) evidencePriori SG, Blomström-Lundqvist C. 2015 European Society of Cardiology Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death summarized by co-chairs. European heart journal. 2015 Nov 1:36(41):2757-9. doi: 10.1093/eurheartj/ehv445. Epub [PubMed PMID: 26745817]
Lip GY, Heinzel FR, Gaita F, Juanatey JR, Le Heuzey JY, Potpara T, Svendsen JH, Vos MA, Anker SD, Coats AJ, Haverkamp W, Manolis AS, Chung MK, Sanders P, Pieske B, Gorenek B, Lane D, Boriani G, Linde C, Hindricks G, Tsutsui H, Homma S, Brownstein S, Nielsen JC, Lainscak M, Crespo-Leiro M, Piepoli M, Seferovic P, Savelieva I. European Heart Rhythm Association/Heart Failure Association joint consensus document on arrhythmias in heart failure, endorsed by the Heart Rhythm Society and the Asia Pacific Heart Rhythm Society. Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. 2016 Jan:18(1):12-36. doi: 10.1093/europace/euv191. Epub 2015 Aug 21 [PubMed PMID: 26297713]
Level 3 (low-level) evidenceAbdelhamid MA, Samir R. Reversal of premature ventricular complexes induced cardiomyopathy. Influence of concomitant structural heart disease. Indian heart journal. 2018 May-Jun:70(3):410-415. doi: 10.1016/j.ihj.2017.08.025. Epub 2017 Sep 1 [PubMed PMID: 29961459]
Belhassen B. Radiofrequency ablation of "benign" right ventricular outflow tract extrasystoles: a therapy that has found its disease? Journal of the American College of Cardiology. 2005 Apr 19:45(8):1266-8 [PubMed PMID: 15837260]
Penela D, Fernández-Armenta J, Aguinaga L, Tercedor L, Ordoñez A, Bisbal F, Acosta J, Rossi L, Borras R, Doltra A, Ortiz-Pérez JT, Bosch X, Perea RJ, Prat-González S, Soto-Iglesias D, Tolosana JM, Vassanelli F, Cabrera M, Linhart M, Martinez M, Mont L, Berruezo A. Clinical recognition of pure premature ventricular complex-induced cardiomyopathy at presentation. Heart rhythm. 2017 Dec:14(12):1864-1870. doi: 10.1016/j.hrthm.2017.07.025. Epub 2017 Jul 27 [PubMed PMID: 28756100]
Abdalla IS, Prineas RJ, Neaton JD, Jacobs DR Jr, Crow RS. Relation between ventricular premature complexes and sudden cardiac death in apparently healthy men. The American journal of cardiology. 1987 Nov 1:60(13):1036-42 [PubMed PMID: 3673904]
Bikkina M, Larson MG, Levy D. Prognostic implications of asymptomatic ventricular arrhythmias: the Framingham Heart Study. Annals of internal medicine. 1992 Dec 15:117(12):990-6 [PubMed PMID: 1280018]
Panizo JG, Barra S, Mellor G, Heck P, Agarwal S. Premature Ventricular Complex-induced Cardiomyopathy. Arrhythmia & electrophysiology review. 2018 Jun:7(2):128-134. doi: 10.15420/aer.2018.23.2. Epub [PubMed PMID: 29967685]
Massing MW, Simpson RJ Jr, Rautaharju PM, Schreiner PJ, Crow R, Heiss G. Usefulness of ventricular premature complexes to predict coronary heart disease events and mortality (from the Atherosclerosis Risk In Communities cohort). The American journal of cardiology. 2006 Dec 15:98(12):1609-12 [PubMed PMID: 17145219]
Lee V, Perera D, Lambiase P. Prognostic significance of exercise-induced premature ventricular complexes: a systematic review and meta-analysis of observational studies. Heart Asia. 2017:9(1):14-24. doi: 10.1136/heartasia-2016-010854. Epub 2017 Jan 4 [PubMed PMID: 28123456]
Level 1 (high-level) evidenceTaylor RS, Sagar VA, Davies EJ, Briscoe S, Coats AJ, Dalal H, Lough F, Rees K, Singh S. Exercise-based rehabilitation for heart failure. The Cochrane database of systematic reviews. 2014 Apr 27:2014(4):CD003331. doi: 10.1002/14651858.CD003331.pub4. Epub 2014 Apr 27 [PubMed PMID: 24771460]
Level 1 (high-level) evidenceBäck M, Öberg B, Krevers B. Important aspects in relation to patients' attendance at exercise-based cardiac rehabilitation - facilitators, barriers and physiotherapist's role: a qualitative study. BMC cardiovascular disorders. 2017 Mar 14:17(1):77. doi: 10.1186/s12872-017-0512-7. Epub 2017 Mar 14 [PubMed PMID: 28288580]
Level 2 (mid-level) evidenceBan JE, Park HC, Park JS, Nagamoto Y, Choi JI, Lim HE, Park SW, Kim YH. Electrocardiographic and electrophysiological characteristics of premature ventricular complexes associated with left ventricular dysfunction in patients without structural heart disease. Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. 2013 May:15(5):735-41. doi: 10.1093/europace/eus371. Epub 2012 Nov 29 [PubMed PMID: 23194696]
Lin CY, Chang SL, Lin YJ, Chen YY, Lo LW, Hu YF, Tuan TC, Chao TF, Chung FP, Liao JN, Chang YT, Lin CH, Walia R, Te ALD, Yamada S, Chiou CW, Tsao HM, Chen SA. An observational study on the effect of premature ventricular complex burden on long-term outcome. Medicine. 2017 Jan:96(1):e5476. doi: 10.1097/MD.0000000000005476. Epub [PubMed PMID: 28072689]
Level 2 (mid-level) evidence